MOTS-c Peptide Dosage: The Ultimate Research Guide
How do you set a mots-c peptide dosage when the published map is incomplete and most online advice skips the hardest part, translating early research into a defensible protocol?
That’s the central problem with MOTS-c. It’s a mitochondrial-derived peptide with growing interest in metabolism and exercise-related research, but the evidence base is uneven. Preclinical work offers useful anchors. Human data are narrower, and established clinical dosing standards don’t exist.
A careful researcher should treat dosage as a process, not a fixed number copied from a forum post. Mechanism matters. Route matters. Species differences matter. Preparation quality matters. Above all, MOTS-c remains a research compound, not a drug with FDA-established human dosing.
Table of Contents
- Introduction What is MOTS-c and Why is Dosage a Challenge
- The Core Mechanism How MOTS-c Influences Metabolism
- Key Factors Influencing MOTS-c Dosage Selection
- A Review of Preclinical MOTS-c Dosing Protocols
- How to Calculate MOTS-c Dosage for Laboratory Research
- Reconstitution Administration and Protocol Cycling
- Safety Ethics and the Regulatory Landscape
- Conclusion Navigating MOTS-c Dosage with Precision
Introduction What is MOTS-c and Why is Dosage a Challenge
MOTS-c stands out because it’s discussed as a mitochondrial signaling peptide rather than a conventional small molecule. That unusual biology is part of the appeal. It’s also why dosage discussions get messy fast.
Researchers often want a clean answer such as “use X mg” or “dose by body weight.” The current literature doesn’t support that level of certainty. Instead, it supports a structured approach that starts with mechanism, checks the preclinical protocol, and then narrows the dosing hypothesis based on route, subject type, and study objective.
Two mistakes cause most confusion. The first is treating animal mg/kg data as if it can be copied directly into human-oriented protocols. The second is assuming that common wellness-style schedules are the same thing as validated clinical standards. They aren’t.
Working principle: with MOTS-c, dosage selection is part pharmacology, part study design, and part quality control.
That’s why a useful guide to mots-c peptide dosage has to focus less on slogans and more on decision logic.
The Core Mechanism How MOTS-c Influences Metabolism

What biological event is a MOTS-c dose trying to produce?
That question matters because MOTS-c is studied less like a simple replacement compound and more like a signaling cue. In preclinical papers, the peptide is commonly discussed in relation to AMPK, a central regulator of cellular energy balance. For researchers, the practical implication is clear. Dose selection is tied to target engagement, not just to the amount placed into a syringe.
Why AMPK matters for dose selection
AMPK functions like a cellular fuel gauge. When energy availability shifts, AMPK helps redirect metabolism toward restoring balance. That is why MOTS-c attracts attention in metabolic research. Investigators are not only asking whether the peptide is present. They are asking whether it changes energy handling in a measurable way.
This distinction helps prevent a common error in dosage discussions. A higher nominal dose does not automatically mean a more informative experiment. If the study goal is to examine glucose handling, exercise adaptation, or mitochondrial stress signaling, the useful dose is the one that produces interpretable pathway activity under the chosen model conditions.
Human evidence remains limited, and established clinical dosing standards do not exist for routine use. Much of the mechanistic framework still comes from animal and cell research. That makes MOTS-c dosage a research design problem before it becomes a calculation problem.
Mechanism first, then quantity
A simple analogy helps. Setting a MOTS-c dose is closer to tuning a stimulus in a signaling experiment than to filling a reservoir to a fixed level. Researchers are trying to produce a biological response window. Too little exposure may miss the pathway. Too much may change the system in ways that no longer match the original question.
That is one reason route and schedule matter so much in metabolism studies. A peptide delivered by one route can reach circulation at a different rate and extent than the same nominal amount delivered by another route. Two protocols can list similar numbers and still represent different exposure conditions.
Why route changes the interpretation
Route should be treated as part of the dosing hypothesis, not as a footnote. In preclinical work, intraperitoneal administration is often used because it provides controlled delivery in animal models. In human-oriented research settings, subcutaneous administration is more commonly discussed for practical reasons. Those routes are not interchangeable.
| Research context | Common route described | Why it matters |
|---|---|---|
| Preclinical animal work | Intraperitoneal | Often used for controlled delivery in animal models |
| Human-oriented research protocols | Subcutaneous | More practical, but not directly equivalent to animal intraperitoneal dosing |
The same principle applies to frequency. Daily exposure in an animal protocol is part of the experimental model, not a universal template. Once route, frequency, and species change, the meaning of the original dose changes with them.
Route is part of the dose. If route changes, the comparison changes too.
For MOTS-c, the safest way to read the mechanism literature is to treat it as a map of possible biological effects, not as a ready-made human protocol. That process-oriented view helps bridge two bad extremes. One is reading papers so narrowly that dose design becomes impossible. The other is copying anecdotal schedules without respecting how preclinical signaling data were generated.
Key Factors Influencing MOTS-c Dosage Selection

What determines a reasonable mots-c peptide dosage in research. The answer is rarely a single number. It is a chain of decisions that starts with the study objective, then narrows through biology, protocol design, and the limits of the available evidence.
That process matters because MOTS-c sits in an awkward translational space. Preclinical findings give researchers a starting framework, but they do not provide an established human dosing standard. Anecdotal schedules often skip that distinction. Careful protocol design cannot.
The research question sets the boundaries
Dose selection should begin with the endpoint being tested. A study designed to examine metabolic markers is asking a different question than one focused on exercise performance, body composition, or early tolerability observations. If the question changes, the dose rationale often changes with it.
A useful way to frame this is to treat dosage like the exposure setting on a microscope. The setting depends on what the researcher is trying to see. Higher exposure is not automatically better, and lower exposure is not automatically safer in an interpretable way. The selected dose has to match the signal under investigation.
Earlier evidence discussed in this article shows why caution is necessary. Human analog work has been explored, and preclinical animal studies have also reported measurable effects, but those findings do not collapse into one universal protocol. They define a range of research considerations, not a ready-made rule.
Biological variables change what a dose means
Two studies can use the same nominal amount and still be probing different biological conditions. That is one reason translation from animal data to human-oriented research is difficult.
Several variables deserve attention:
- Sex-related response differences: Prior reviews have raised the possibility that men and women may not respond identically, so sex should be specified in protocol planning rather than treated as background detail.
- Species-specific metabolism: Mouse and human metabolic systems overlap in broad pathways, but they differ in rate, distribution, and downstream response. A dose that produces a clear murine signal may still leave uncertainty about relevance outside that model.
- Baseline physiological state: Lean animals, obese animals, trained subjects, sedentary subjects, and metabolically impaired models do not represent the same starting condition. The same dose can produce different observed effects across those settings.
- Target endpoint: A study looking for changes in glucose handling may justify a different exposure strategy than one focused on endurance or body composition.
- Conservative escalation logic: In exploratory work, researchers often begin with a defensible low-end assumption and revise only after confirming that the protocol is functioning as intended.
Those factors are easy to list and harder to apply. The practical question is not, “What is the MOTS-c dose?” The practical question is, “What dose range is defensible for this model, this endpoint, and this study design?”
This short explainer can help frame those design choices before drafting a protocol:
A careful researcher treats dose selection as a justification exercise. Each variable acts like a conversion step in a calculation. Skip one, and the final number may look precise while resting on weak assumptions.
A Review of Preclinical MOTS-c Dosing Protocols

Published animal studies give something more useful than internet dose anecdotes. They give a protocol shape. That matters because MOTS-c discussions often jump from “it affected metabolism in mice” to unsupported dosing claims. The preclinical record is narrower and more disciplined than that.
What the animal literature actually gives you
A recurring pattern in the mouse literature is straightforward: defined doses, a defined route, and a defined study window. In diet-induced obesity models, investigators have used intraperitoneal MOTS-c over a multi-week period, including protocols built around 5 mg/kg and 15 mg/kg for 21 days, with stronger metabolic and performance-related effects reported at the higher end of that range, as noted earlier.
That type of paper helps in one specific way. It shows how researchers paired dose with an experimental question. The aim was not merely to “administer MOTS-c.” The aim was to test whether a chosen exposure level could shift outcomes such as body composition, glucose-related measures, or exercise capacity within a controlled model.
A dosing protocol works like a lab assay. Its meaning depends on the full setup. Remove the species, route, disease model, and observation window, and the number by itself loses much of its value.
How to read those protocols responsibly
Preclinical dosing papers are best treated as boundary markers for research design.
They can inform:
- Study planning in animal models with similar metabolic endpoints
- Range-finding decisions before a full efficacy experiment
- Mechanistic work where the goal is to link exposure to downstream signaling or performance outcomes
- Protocol comparison across obesity, exercise, or age-related models
They do not establish:
- Medical dosing guidance
- A validated human dosing standard
- Interchangeability across administration routes
- Proof that commercially available MOTS-c products match published research material
That distinction is easy to miss. A mouse protocol can justify another mouse experiment. It does not automatically justify a human schedule, a different route, or a different formulation.
The safest way to use this literature is to ask a narrow question: under what exact conditions did a given dose produce an observable effect? Once that question is answered, the paper becomes useful. Without that discipline, preclinical numbers get copied out of context and start to look more certain than they are.
How to Calculate MOTS-c Dosage for Laboratory Research
Calculation is where a lot of readers get stuck. The arithmetic is simple. The hard part is knowing which number belongs in the equation and which number is just a protocol reference from a different context.
Start with the protocol not the vial
A vial label doesn’t tell you the correct dose. Your protocol does.
For laboratory work, dose calculation usually moves through this order:
-
Define the target dose
Use the study protocol first. If the design is based on an animal model, that may be an mg/kg target. If the design follows a published human-oriented research protocol, it may be a fixed mg-per-injection schedule. -
Determine subject body weight if relevant
For mg/kg work, multiply body weight by the target dose. -
Calculate the total amount needed per administration
Example formula:
Dose required = body weight × target mg/kg -
Only then calculate injection volume
Volume depends on the concentration after reconstitution.
A common mistake is to start by asking how many units to draw. That question comes last.
Turn dose into volume after reconstitution
Once a peptide is reconstituted, the vial has a concentration. Volume is calculated from that concentration.
Example using simple lab math:
- If a vial contains 10 mg of peptide reconstituted with 1 mL diluent, the concentration is 10 mg/mL
- If the protocol requires 5 mg, then the needed volume is 0.5 mL
Formula:
Volume to draw = required dose ÷ concentration
That same logic applies whether the protocol is mg/kg based or fixed-dose based. The protocol defines the amount. The concentration defines the volume.
Commonly referenced research-style protocols have explored starting doses of 5 mg per injection, with weekly totals of 10 to 15 mg/week divided into 2 to 3 subcutaneous injections. More specialized protocols have used 5 mg every 5 days for a limited cycle (summary of commonly referenced MOTS-c dosing protocols).
A compact workflow helps:
| Step | Question | Output |
|---|---|---|
| 1 | What dose does the protocol require? | mg or mg/kg |
| 2 | Is body weight part of the design? | total mg needed |
| 3 | What is the final concentration after reconstitution? | mg/mL |
| 4 | How much volume delivers that amount? | mL to administer |
Calculation rule: never convert vial volume until the protocol dose is fixed in mg.
This sounds basic, but it’s the difference between a reproducible protocol and guesswork.
Reconstitution Administration and Protocol Cycling
What turns a mathematically correct dose into a misleading experiment? In peptide work, the answer is often the process around the dose. With MOTS-c, reconstitution method, administration timing, and cycle structure can all change how interpretable the study remains.
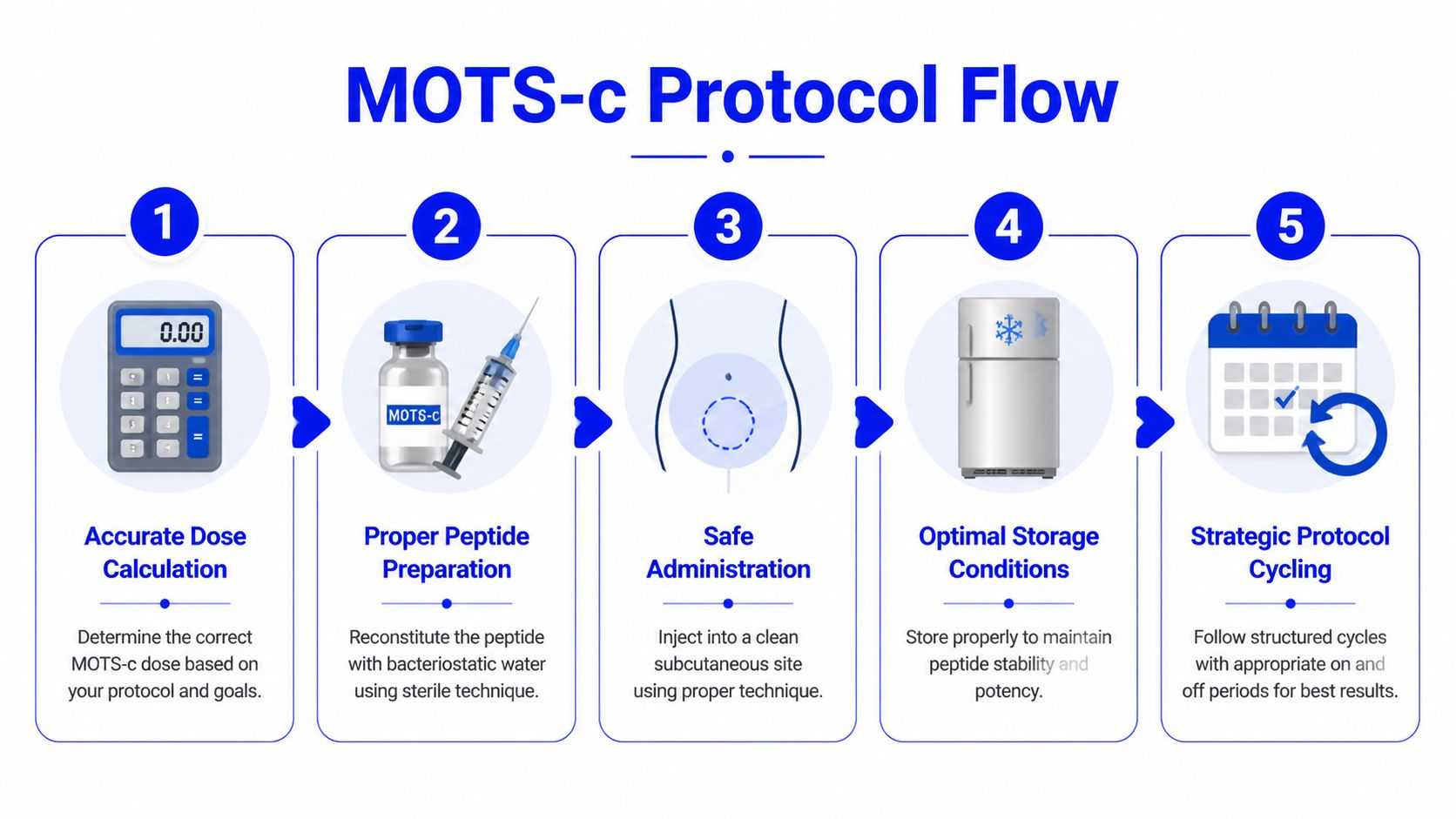
Preparation errors can distort the study
Lyophilized peptides are chemically simple on paper and procedurally sensitive at the bench. A vial can contain the intended amount of MOTS-c and still yield inconsistent exposure if the peptide is mishandled during reconstitution or storage. For that reason, preparation should be treated as part of the dosing protocol, not as a routine setup step.
Gentle handling matters. Vigorous shaking can increase foaming, surface stress, and other conditions that may reduce peptide integrity. The practical rule is straightforward. Add diluent carefully, let the powder dissolve, and mix with slow inversion or a light swirl rather than agitation.
Small deviations create large interpretive problems later. If one animal cohort receives material prepared with a different technique, or if storage records are incomplete, the study can no longer cleanly separate a dosing effect from a handling effect.
A defensible preparation routine usually includes:
- Sterile setup: use a clean workspace, sterile tools, and lot-specific labeling before reconstitution begins
- Controlled mixing: dissolve the peptide gently and avoid aggressive shaking
- Exact documentation: record the diluent type, final volume, date, time, and operator
- Storage tracking: tie temperature and storage duration to the same batch record used for dosing
Administration timing and cycle design
Administration schedules need the same level of discipline. A dose is only one dimension of exposure. Frequency, time of day, total cycle length, and washout period all shape what the experiment is testing.
Many informal MOTS-c discussions become unhelpful; they jump from a vial strength to a repeated-use pattern without explaining the logic in between. A better research approach starts with the study question. If the goal is to examine metabolic adaptation, the protocol should define in advance how long exposure continues, whether administration time stays fixed, and what evidence would justify a washout period.
Some research-oriented discussions have described multi-week administration blocks followed by a washout interval, with dosing time kept consistent to reduce circadian noise in the data (protocol discussion of timing and cycling). That should be read as protocol design context, not as an established human standard. MOTS-c does not have a settled clinical dosing framework, so timing and cycling choices belong inside a clearly defined research plan.
A practical checklist helps keep that plan reproducible:
- Define the full cycle before the first administration so schedule changes do not creep in mid-study
- Set washout criteria prospectively rather than after response patterns become obvious
- Keep time-of-day consistent if the protocol assumes circadian relevance
- Separate dosing logs from assay outcome logs so preparation or timing errors can be traced without rewriting the record
Cycling serves a methodological purpose. It helps the investigator examine adaptation, recovery, and carryover effects with fewer hidden variables.
For labs sourcing research material, one factual option in this category is Peptide Warehouse USA, which lists MOTS-C 10 mg vials for laboratory and analytical use and provides batch documentation such as COAs and related testing records through its research-supply framework.
Safety Ethics and the Regulatory Landscape
What does a careful MOTS-c dosage discussion look like when the compound is still defined more clearly in preclinical research than in routine human medicine? It starts with a simple point. Dose calculations are only one part of study quality. Material identity, documentation, ethics review, and accurate claims matter just as much.
That matters because MOTS-c sits in an awkward middle ground. There is enough published research to justify scientific interest, but not enough to support casual translation into human use. In other words, a clean spreadsheet cannot rescue a poorly sourced peptide or an overstated conclusion. If the input material is uncertain, the dose is uncertain in a practical sense, even if the arithmetic is correct.
What responsible sourcing looks like
A MOTS-c protocol begins with chain-of-custody questions, not with a syringe volume or mg/kg conversion. The comparison is straightforward. Measuring dose without confirming identity is like calibrating an instrument before checking whether the sample in the tube is the one named on the label.
Researchers should be able to verify several basic points before treating any result as interpretable:
- Is there a third-party COA tied to the exact lot being used?
- Are purity, identity, and batch traceability documented clearly enough for later audit or replication?
- Are microbial or endotoxin records available when the research setting makes them relevant?
- Can the supplier account for storage conditions, packaging, and labeling in a way that matches the stated material?
- Is the product sold under research-only compliance rules?
Missing records create a specific problem. They make it hard to tell whether an observed effect came from the protocol, the peptide itself, degradation during handling, or contamination. At that point, dosage precision becomes less informative because the test article is no longer well characterized.
Why regulatory caution matters
The current evidence base does not support casual use or casual language. Public-facing discussions can blur an important distinction. A peptide may be biologically interesting and commercially available while still lacking regulatory approval for standard therapeutic use.
That distinction should shape how MOTS-c is discussed in papers, lab notes, product pages, and conference conversations. The Alzheimer’s Drug Discovery Foundation has noted the absence of an established clinical dosing standard, and human discussion has centered on an analog rather than a broadly approved therapeutic protocol. That is not a minor footnote. It changes what can be claimed responsibly.
A few guardrails help keep the process honest:
- MOTS-c is not an FDA-approved standard therapy
- Preclinical findings should not be rewritten as medical advice
- Commercial sale does not equal regulatory approval
- Unknowns remain around long-term exposure, context-specific risk, and protocol selection
A responsible researcher treats uncertainty as part of the result, not as permission to fill gaps with confidence.
Ethics enters at the point where enthusiasm begins to outrun evidence. Good practice slows that process down. It records assumptions before the study starts, separates preclinical reasoning from human claims, and keeps the compound inside a research-only context unless stronger evidence and formal oversight justify something more.
Conclusion Navigating MOTS-c Dosage with Precision
The hardest part of mots-c peptide dosage isn’t the math. It’s respecting the boundaries of the evidence.
The current literature gives a useful starting framework. It links MOTS-c to AMPK-centered metabolic effects, provides specific animal dosing models, and shows that human analog research has begun. What it does not give is a universal, FDA-established dosing standard for routine human use.
That leaves researchers with a clear responsibility. Build the protocol from the mechanism outward. Keep preclinical and human-oriented evidence separate. Treat route, reconstitution, timing, and cycling as core variables, not side notes. Verify material quality before drawing conclusions from any result.
A precise MOTS-c protocol is possible when each step is documented and justified. A careless one can look scientific while resting on unsupported assumptions.
If you’re comparing research peptide suppliers, looking for lot documentation, or reviewing available MOTS-c formats for laboratory use, you can learn more at Peptide Warehouse USA and explore options that fit a research-only workflow.






Leave a comment