The Truth About tb 500 peptide benefits
If you’re evaluating tb 500 peptide benefits for a lab, you’re probably sorting through two very different kinds of content. One side treats TB-500 like a miracle compound. The other side dismisses it because the human evidence is thin. Neither view is useful if your job is to design clean studies, choose appropriate comparators, and source material that won’t compromise the data.
TB-500 is worth attention because the preclinical signal is real, especially in models of tissue repair, inflammation modulation, and cellular migration. But the enthusiasm needs boundaries. Most of what looks promising comes from animal and mechanistic research, not established human protocols.
This article looks at TB-500 the way a research scientist or experienced lab manager should. What it is. Why it’s studied. Which findings are supported in preclinical work. Where the safety and dosing gaps still are. And why compound quality matters if you’re trying to generate results that hold up under scrutiny.
Table of Contents
- Introduction A Primer on TB-500 for Research
- What Is TB-500 and Why Is It Studied
- The Core Mechanism How TB-500 Influences Cellular Repair
- Investigated TB-500 Peptide Benefits in Preclinical Models
- How TB-500 Compares to BPC-157 in Research Applications
- Navigating Safety and Dosing in a Research Context
- Ensuring Research Integrity with High-Purity TB-500
- Conclusion The Future of TB-500 in Regenerative Research
Introduction A Primer on TB-500 for Research
TB-500 keeps showing up in conversations about repair-focused compounds for one reason. It sits at the intersection of cell migration, tissue remodeling, and inflammation control, which makes it relevant to many preclinical models.
In plain terms, TB-500 is a synthetic peptide associated with Thymosin Beta-4 biology. Researchers study it because that biology appears closely tied to how cells move, reorganize structural proteins, and respond to injury. Those are not niche questions. They matter in wound models, soft tissue studies, cardiovascular injury work, and broader regenerative research.
The phrase tb 500 peptide benefits is often used loosely online, but in a serious setting the better question is narrower. Which observed effects are supported in preclinical work, and which claims have moved faster than the evidence?
Practical rule: Treat TB-500 as a research tool with a defined mechanistic rationale, not as a catch-all recovery claim.
That distinction changes how you read the literature, how you design experiments, and how much weight you give to outcome data from different model types.
What Is TB-500 and Why Is It Studied
A common lab scenario is straightforward. A team is screening compounds for an injury model, and most candidates arrive wrapped in broad recovery claims with little mechanistic discipline. TB-500 keeps drawing attention because the question is narrower and more testable. Researchers study it as a synthetic peptide associated with Thymosin Beta-4 biology, where the interest centers on cell movement, structural organization, and tissue response after injury.
That distinction matters.
In preclinical work, TB-500 is not interesting because it sits inside a vague wellness category. It is interesting because it points to a defined area of biology. The compound is typically discussed in relation to actin-regulated processes, which gives investigators a plausible reason to test it in wound, musculoskeletal, vascular, and other repair-focused models. For a research group, that is a better starting point than generalized claims about faster recovery.
The practical appeal is methodological as much as biological. If a compound may affect how cells migrate, spread, and reorganize after damage, it becomes relevant to experiments where timing, tissue architecture, and repair quality can be measured directly. That makes TB-500 easier to place in a study design than many peptides that are marketed first and explained later.
Researchers usually return to the same set of preclinical questions:
- Cell migration: Whether repair-relevant cells reach the injury site more efficiently
- Tissue remodeling: Whether damaged structures reorganize in a more favorable pattern
- Angiogenic activity: Whether local vascular support changes during recovery
- Inflammatory signaling: Whether the injury environment shifts in ways that may support repair
Those are research questions, not confirmed clinical outcomes.
Another point is easy to miss in online discussions. TB-500 is often treated as interchangeable with Thymosin Beta-4 itself, but careful readers should avoid collapsing those terms. The literature and product descriptions do not always use them with the same level of precision, and that creates problems when comparing studies, interpreting mechanisms, or sourcing material for experimental use. For anyone trying to evaluate the evidence seriously, compound identity and formulation are part of the story, not a footnote.
The reason TB-500 remains under active discussion is simple. It has a coherent preclinical rationale, and that is more than can be said for many compounds in this category. The gap is that mechanistic plausibility does not settle efficacy, optimal use conditions, or translatability. Those questions still depend on better-designed studies and well-characterized, high-purity material.
The Core Mechanism How TB-500 Influences Cellular Repair
A common lab scenario is straightforward. A compound shows up in wound and soft-tissue studies, the readouts look promising, and then the mechanism gets flattened into a vague claim about “healing support.” TB-500 deserves a tighter explanation than that.
Actin regulation is the center of the story
The most credible mechanistic frame for TB-500 centers on actin dynamics. Actin is part of the cellular scaffold, but in repair biology it is also a movement system. Cells involved in regeneration need to change shape, migrate into damaged tissue, attach, detach, and reorganize local structure. If a peptide influences those processes, it can affect repair long before anyone talks about a final tissue outcome.
TB-500 has been described as influencing repair through effects linked to actin regulation and cell migration, as summarized in this review discussing TB-500’s mechanism and metabolite question. That framing fits why the compound keeps appearing in discussions of skin, muscle, tendon, ligament, and wound models. It does not prove broad efficacy across all of those settings. It does provide a coherent reason to study them.
From a research perspective, the mechanism points to a specific set of downstream effects:
- Altered migration of repair-relevant cells into injured tissue
- Changes in how damaged tissue is reorganized during recovery
- Support for vascular responses that can sustain repair
- Shifts in local inflammatory signaling that may favor restoration over persistence of injury
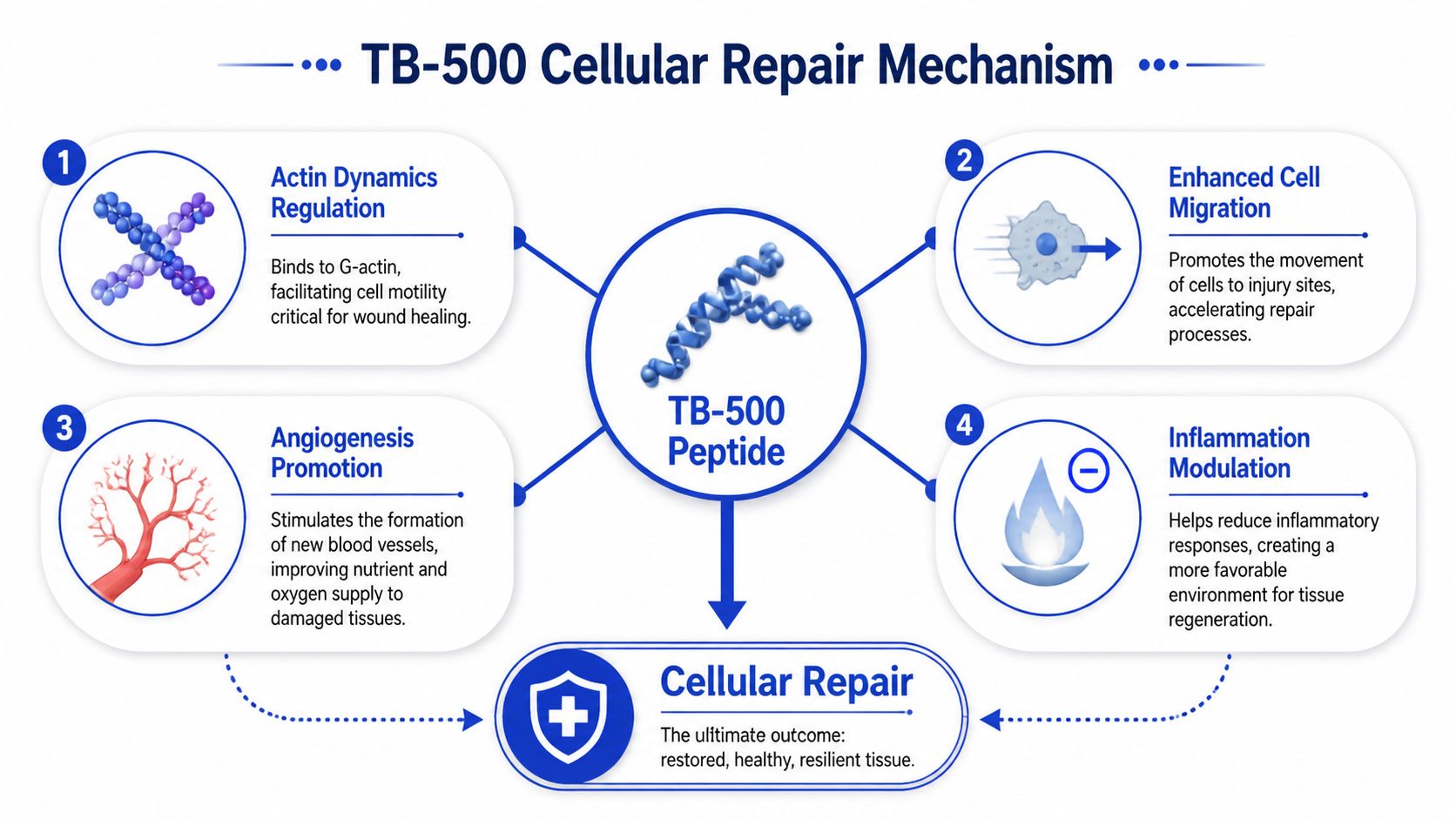
Those are mechanistic hypotheses with preclinical support, not interchangeable endpoints. In practice, that distinction matters. A model can show improved cell motility without showing stronger healed tissue, and a histology result can look favorable while functional recovery remains unchanged.
The metabolite question matters
A serious reading of TB-500 also has to account for the metabolite Ac-LKKTE. Recent discussion in the field has raised the possibility that part of the observed biological activity may depend on metabolic processing rather than the parent peptide alone, as noted earlier in the same review.
That changes how experiments should be designed. If conversion contributes to activity, then nominal dose is only one variable. Researchers also need to ask whether the model can generate the relevant metabolite, whether the assay window captures that conversion, and whether an in vitro system is too reductive for the question being asked.
This is one of the main trade-offs in TB-500 work. A simplified system can isolate a clean mechanistic signal, but it may miss metabolism-dependent effects. A more physiologic model may be closer to the biology of interest, but it also introduces more confounders.
The sourcing implications are equally important. If metabolism is part of the mechanism, then purity, sequence identity, and analytical documentation matter because they affect interpretation, not just procurement.
Lab takeaway: For TB-500, the parent peptide may be only one part of the signal. Metabolic conversion can change what your model is measuring.
That is why broad claims about “systemic healing” add little value. The better research question is narrower. Which repair pathway is being influenced, under what conditions, and with what evidence that the observed effect belongs to TB-500, its metabolites, or both?
Investigated TB-500 Peptide Benefits in Preclinical Models
A familiar lab scenario is a repair model that closes on schedule but produces disorganized tissue, persistent inflammation, or poor remodeling. That is the context in which TB-500 becomes interesting. The serious case for tb 500 peptide benefits comes from preclinical work that examines repair quality, migration-dependent healing, and tissue response under controlled conditions.

Accelerated healing and tissue repair
The strongest signal remains wound healing. A single medical evidence review summarizing TB-500 wound-healing studies describes faster cutaneous wound closure in animal models, shorter healing intervals in treated groups, improved tensile strength of repaired tissue, and favorable results in chronic wound settings.
Those findings fit the biology under investigation. If a compound affects actin dynamics and cell migration, better re-epithelialization and more coordinated repair are plausible readouts.
The trade-off is interpretation. Review-level summaries are useful for seeing the direction of the literature, but they compress differences in model design, dosing schedules, peptide handling, and outcome definitions. A lab planning replication should treat the wound signal as promising, then go back to the underlying animal studies and ask a narrower question. Was the main effect on closure speed, tissue strength, inflammatory tone, or all three?
Inflammation modulation and scar-related observations
Some of the more interesting preclinical observations are not about speed at all. They concern the repair environment.
The same review notes reduced pro-inflammatory signaling, including lower TNF-α and NF-κB activity in certain models, alongside observations consistent with less pronounced pathologic scarring. That pattern matters because faster closure alone can be misleading. A wound that closes quickly but remodels poorly is not a clear success in most research programs.
For experimental planning, it helps to separate the endpoints:
| Research question | Why TB-500 is relevant |
|---|---|
| Does tissue close faster? | Migration-dependent repair may improve wound coverage |
| Does tissue remodel with better structure? | Preclinical tensile strength and scar-related findings suggest this is worth testing |
| Is the inflammatory response altered? | Cytokine and signaling changes indicate a potentially different repair environment |
A short explainer can help contextualize the broader discussion:
Cardiac and broader regenerative signals
Interest in TB-500 extends beyond skin and soft tissue models. Preclinical literature has explored cardiac injury, vascular response, and neural repair, with reports of improved recovery-related markers in some settings. As noted earlier in the article, these broader regenerative signals are one reason the peptide continues to attract attention in research circles.
This is also where the hype tends to outrun the evidence. Multi-tissue activity in animal work does not establish a universal repair agent, and it does not remove the need for model-specific validation. Cardiac ischemia, tendon injury, and neural trauma do not share the same constraints, even if they all involve migration, remodeling, or cytoskeletal reorganization.
The practical reading of the literature is narrower and more useful. TB-500 appears most relevant in preclinical systems where the endpoint depends on coordinated cell movement, tissue organization, and recovery from structural disruption. Future studies using high-purity material, tight analytical controls, and metabolite-aware designs should clarify which effects belong to the parent peptide, which depend on downstream processing, and which are artifacts of model choice.
How TB-500 Compares to BPC-157 in Research Applications
TB-500 and BPC-157 are often mentioned in the same breath, which creates confusion. For research planning, they shouldn’t be treated as interchangeable.

Different mechanisms, different experimental logic
A useful distinction comes from this discussion of TB-500 versus BPC-157 mechanisms in peptide research. TB-500 specifically targets actin, which is central to cellular movement. BPC-157 is described there as operating through angiogenesis and fibroblast migration.
That difference changes the experimental logic.
If your model focuses on cell motility, cytoskeletal dynamics, or systemic repair signaling, TB-500 may be the cleaner mechanistic fit. If the model leans toward vascular support and fibroblast-driven local repair, BPC-157 may match the question more directly.
Neither choice should be made based on online reputation alone. The mechanism should match the endpoint.
A practical selection framework
When labs compare the two, I use a simple decision screen:
- Choose TB-500-focused work when the central question involves actin regulation, distributed tissue response, or migration-dependent repair.
- Choose BPC-157-focused work when the priority is local repair signaling, angiogenic support, or fibroblast-related healing pathways.
- Study both cautiously when the goal is comparison or synergy, but only if the design can separate pathway effects rather than blending them into one vague “recovery” outcome.
Mechanistic clarity matters more than popularity. If you can’t explain why a peptide belongs in the model, you probably shouldn’t build the experiment around it.
That’s where a lot of weak peptide content fails. It treats TB-500 and BPC-157 as branding categories, not as distinct research tools.
Navigating Safety and Dosing in a Research Context
The most responsible thing to say about TB-500 safety is also the least exciting. The long-term human picture is still underdeveloped.
What is known from controlled evidence
A key gap, highlighted in this discussion of TB-500’s limited human safety and dosing evidence, is that there is no established long-term safety data or standardized dosing protocols for human use. The same source notes that a phase 2 trial in severe dry eye demonstrated safety and symptom improvement, but that remains one of the few controlled human studies available.
That matters because a lot of content jumps from preclinical promise to implied clinical confidence. The data don’t justify that leap.
In research settings, the appropriate stance is narrower:
- preclinical safety signals may support further investigation
- isolated human findings show that biological activity in humans is plausible
- standardized human use frameworks still aren’t established
What still shouldn’t be overstated
Many discussions undermine their credibility by presenting animal dose ranges, anecdotal protocols, or informal “stacks” as if they were mature human guidance.
They aren’t.
Even where animal literature reports wide dose tolerance, that information is useful for interpreting preclinical design, not for inferring settled human dosing practice. Differences in metabolism, route, exposure timing, and model relevance can all distort direct comparisons.
A conservative research posture usually means asking questions like these before interpreting a result:
- Is the model capable of generating the active metabolite in a realistic way?
- Are the selected endpoints mechanism-linked, or just broad recovery impressions?
- Does the dosing schedule answer a defined research question?
- Are safety observations being separated from efficacy observations?
Critical caution: A promising preclinical compound can still be poorly characterized for long-term human use. TB-500 fits that description today.
That doesn’t reduce its research value. It defines the limits of what the evidence can support.
Ensuring Research Integrity with High-Purity TB-500
Quality issues can subtly undermine an otherwise well-designed study. With TB-500, that’s especially important because the mechanistic discussion already includes metabolite formation, sequence fidelity, and pathway-specific interpretation.

Purity is not a marketing detail
The available background on TB-500 research quality points to 99.5% purity as a typical benchmark for serious research material, particularly when metabolite profiling and reproducibility matter, as discussed in the earlier mechanistic source and reinforced in the supplier background provided for this article.
Impurities create two problems at once. They can change biological behavior, and they can make a result impossible to interpret. If the endpoint shifts, you won’t know whether the effect came from TB-500, a contaminant, degradation, or inconsistent batch composition.
What careful buyers should verify
For research procurement, the checklist should be straightforward:
- Certificate of Analysis: Confirm identity, stated purity, and batch traceability.
- Third-party documentation: Independent testing reduces the risk of self-reported quality claims.
- Microbial and endotoxin reporting: Important for controlled lab workflows and cleaner interpretation.
- Consistent batch production: Reproducibility depends on lot-to-lot reliability.
- Clear research-use positioning: Serious suppliers make the intended use explicit.
This isn’t glamorous, but it’s where good peptide research starts. A carefully chosen model paired with questionable material still produces questionable work.
Conclusion The Future of TB-500 in Regenerative Research
TB-500 remains one of the more interesting compounds in regenerative research because it offers a clear mechanistic anchor. Its association with actin regulation and cell migration gives researchers something specific to test, not just a vague recovery narrative.
The strongest evidence for tb 500 peptide benefits still comes from preclinical models. Those studies suggest meaningful effects in wound repair, inflammation-related healing conditions, and even cardiac injury research. At the same time, the limits are just as important as the promise. Long-term human safety and standardized human dosing aren’t established, and the metabolite question adds a layer of complexity that researchers shouldn’t ignore.
For serious labs, that combination is familiar. A compelling signal. A real mechanistic rationale. And plenty of unresolved questions that demand better-designed studies rather than louder claims.
Researchers who want reliable inputs for analytical, laboratory, and preclinical work can learn more from Peptide Warehouse USA, a U.S. supplier focused on high-purity research peptides with batch documentation, COAs, and research-only positioning. If you’re comparing sourcing options for TB-500 or related compounds, it’s worth exploring suppliers that prioritize traceability, testing transparency, and consistency before you build the study around the vial.






Leave a comment