Research on MOTS-c Peptide Benefits & Longevity
A mitochondrial peptide that made elderly mice run 2-fold longer and 2.16-fold farther in treadmill tests deserves careful attention, especially when those same research lines also point to improved glucose handling and mitochondrial efficiency in preclinical models (HubMedEd overview of the 2015 findings). That combination is why interest in mots-c peptide benefits keeps growing across metabolism, performance, and longevity research.
The important caveat is just as interesting as the hype. MOTS-c has strong animal and mechanistic data, but clinical translation remains limited, and that gap matters for anyone trying to interpret the science responsibly. If you’re evaluating this peptide for laboratory or preclinical work, the useful question isn’t “is it a miracle?” It’s “what does the evidence support, and where does it stop?”
Table of Contents
- An Introduction to the Mitochondrial Peptide MOTS-c
- What Is MOTS-c and Why Is It Called an ‘Exercise Mimetic’?
- The Molecular Mechanism How MOTS-c Regulates Metabolism
- Primary MOTS-c Peptide Benefits in Metabolic Research
- Exploring Broader Applications in Longevity and Cellular Health
- Key Considerations for Researchers Studying MOTS-c
- Conclusion The Future of MOTS-c in Research
An Introduction to the Mitochondrial Peptide MOTS-c
MOTS-c is a mitochondrial-derived peptide, a class of signaling molecules that originates from the cell’s energy-producing organelles rather than from nuclear DNA. That detail is more than a biochemical curiosity. It places MOTS-c in a signaling system that is closely tied to fuel use, stress adaptation, and metabolic state.
Mitochondria are often described as power plants. A better working analogy is a power plant with sensors, alarms, and outgoing messages. When cellular energy balance shifts, mitochondria do not only make ATP. They also participate in signaling networks that help the cell adjust. MOTS-c appears to be one of those messages, which helps explain why it draws attention in metabolism and aging research.
Researchers study MOTS-c because it connects several questions that are usually examined separately. How does a cell sense energy stress? How are glucose use and fat oxidation re-tuned under that stress? Why do some interventions improve metabolic flexibility in animal models but fail to translate cleanly into human medicine?
That is the scientific appeal here.
Current interest in MOTS-c centers on a few recurring themes:
- Mitochondrial signaling as a regulator of whole-body metabolism
- Glucose control in tissues that are sensitive to energetic demand
- Lipid metabolism during conditions of nutrient excess or metabolic strain
- Cellular stress responses that may influence aging-related decline
- Exercise-like adaptations observed in preclinical models
The preclinical literature is promising, especially in mouse studies of metabolic dysfunction. Investigators have reported improvements in areas such as weight gain under high-fat feeding, glucose homeostasis, exercise capacity, and inflammatory markers. Those findings make MOTS-c a serious experimental topic for labs working on obesity, insulin resistance, sarcopenia, and mitochondrial biology.
At the same time, a careful reading of the field calls for restraint. Preclinical signals do not guarantee clinical usefulness. Peptides can show clean mechanistic effects in controlled animal systems and still run into familiar translational barriers, including dosing, stability, tissue distribution, manufacturing consistency, and the simple fact that human metabolic disease is more heterogeneous than a mouse model.
For a research audience, that balance matters. MOTS-c is neither a fringe curiosity nor an established therapy. It is a biologically plausible mitochondrial signal with substantial preclinical interest and unresolved clinical questions. That is often where the most productive science begins.
What Is MOTS-c and Why Is It Called an ‘Exercise Mimetic’?
MOTS-c is unusually small. It is a 16-amino-acid peptide, yet it has drawn outsized attention because it is encoded within mitochondrial DNA rather than the nuclear genome.
That origin matters. Most signaling molecules discussed in metabolism are framed as messages sent from the cell’s central command structure. MOTS-c is better understood as a signal coming from the energy infrastructure itself. The nucleus works like city hall. Mitochondria work like the power grid. When a peptide is produced from mitochondrial genetic material, researchers immediately ask a different set of questions about fuel sensing, stress adaptation, and metabolic control.
This helps explain why MOTS-c appears so often in discussions of metabolic health and longevity. A mitochondria-derived peptide is well positioned, at least in principle, to influence how cells respond when energy demand rises or nutrient handling becomes inefficient.
A peptide encoded by mitochondrial DNA
The mitochondrial origin of MOTS-c is more than a biochemical curiosity. It gives the peptide a plausible connection to pathways that monitor cellular energy status. In practical terms, that means researchers study it less like a conventional hormone and more like an internal stress and fuel-use signal.
Several features make MOTS-c scientifically interesting:
- Small size: short peptides can act quickly and fit into signaling networks without requiring large, complex structures.
- Mitochondrial encoding: its source links it directly to organelles that govern ATP production and metabolic strain.
- Metabolic relevance: preclinical work connects MOTS-c to glucose handling, lipid use, and adaptive stress responses.
- Translational uncertainty: the strongest evidence still comes from mechanistic studies and animal models rather than established clinical use in humans.
That last point deserves emphasis. MOTS-c has a credible biological rationale and encouraging preclinical data, but those two things do not automatically produce a useful therapeutic agent. Peptides often face familiar barriers such as stability, dosing strategy, tissue exposure, and inter-individual variability in human disease.
Why researchers use the term exercise mimetic
The term exercise mimetic refers to overlap, not equivalence. In the MOTS-c literature, the phrase usually means that the peptide can activate some of the same cellular programs associated with physical training, especially those tied to energy sensing, substrate use, and metabolic adaptation.
A muscle cell during exercise faces a straightforward engineering problem. Energy demand rises rapidly, fuel has to be mobilized, and the cell has to maintain function while limiting metabolic stress. MOTS-c appears to push signaling in a similar direction in some experimental settings. That is why the label persists.
Exercise itself is a whole-body event. It changes blood flow, mechanical loading, neuromuscular recruitment, endocrine signaling, and tissue crosstalk across multiple organs. A peptide may reproduce part of that internal signaling environment, but it does not recreate the full physiology of running, cycling, or resistance training.
For that reason, careful researchers use the phrase with restraint. MOTS-c is best described as a mitochondrial peptide that may induce selected exercise-associated metabolic responses in preclinical models. That is a promising concept for metabolism research. It is also a much narrower claim than saying MOTS-c can substitute for exercise in real-world human physiology.
The Molecular Mechanism How MOTS-c Regulates Metabolism
Across the MOTS-c literature, one signaling node appears again and again: AMPK. That repeated convergence matters because it gives researchers a testable framework for the peptide’s metabolic effects, rather than a vague claim that it “supports mitochondria.”
AMPK is a cellular energy sensor. When ATP supply becomes less favorable relative to energy demand, AMPK shifts cell behavior toward fuel acquisition and efficient ATP production. In practical terms, that means more glucose uptake, greater fatty acid oxidation, and tighter control over energy-intensive processes.
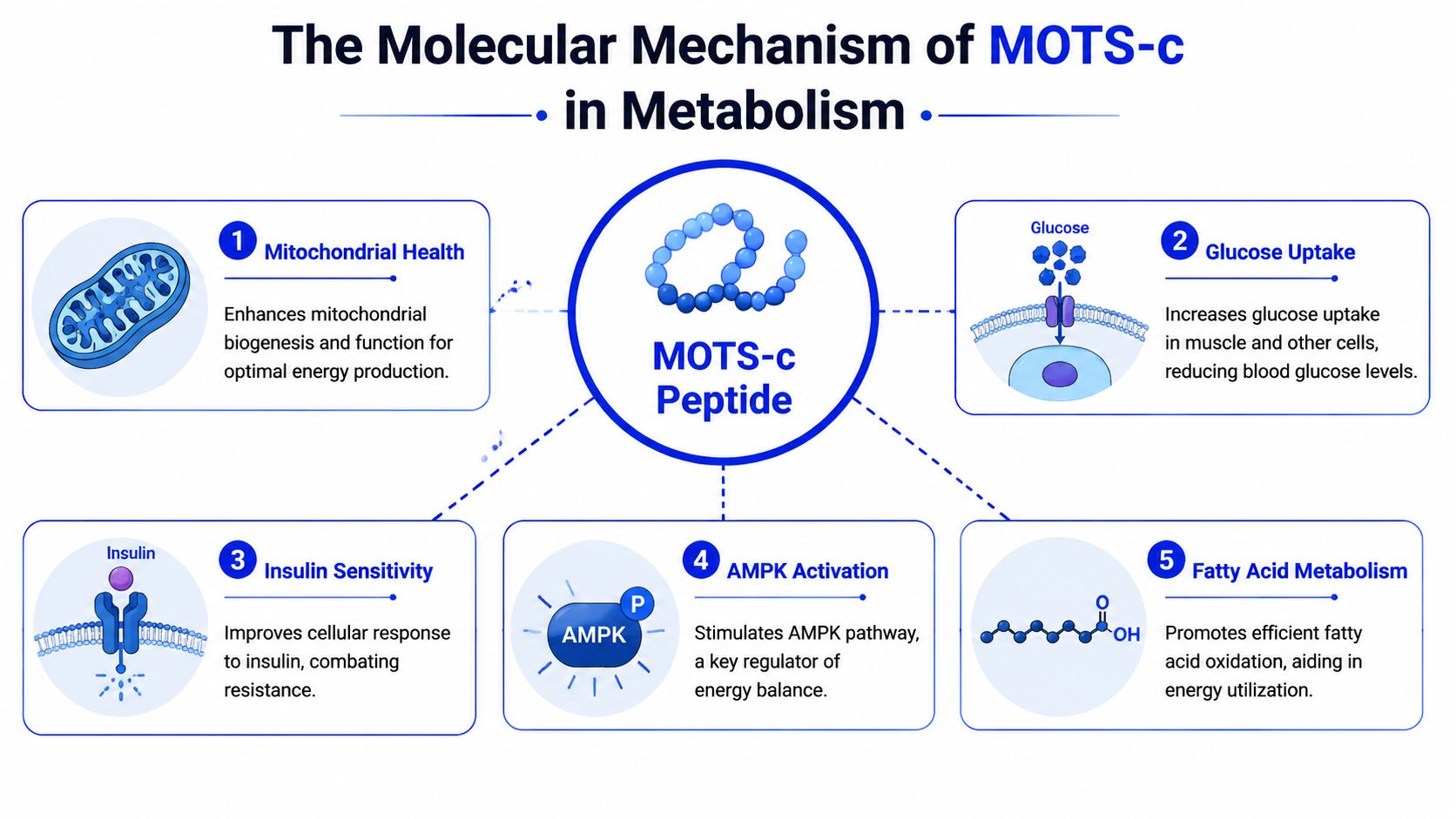
AMPK as the master metabolic switch
The strongest mechanistic interpretation is that MOTS-c shifts cells into an energy-stress response program that depends on AMPK and, in some experimental systems, PGC-1α. That distinction matters. It suggests MOTS-c is working through established metabolic control circuitry, not acting like a nonspecific stimulant.
PGC-1α is especially relevant because it helps coordinate mitochondrial adaptation, oxidative metabolism, and transcriptional responses to energetic demand. If AMPK is the signal that the cell needs to reprioritize fuel use, PGC-1α helps execute part of the longer-term adaptation program. In that sense, the two pathways function less like isolated switches and more like linked layers in a control system.
Preclinical studies support that dependency. When investigators disrupt AMPK or PGC-1α signaling, MOTS-c’s effects on mitochondrial respiration are reduced or lost. For a research audience, that is more informative than broad language about “mitochondrial support” because it identifies which pathways are required for MOTS-c’s effects.
That does not settle the translational question. A pathway can look coherent in cultured cells or mouse muscle and still fail in human disease because of peptide stability, distribution, dose selection, or differences in baseline metabolic state.
For readers who want a broader framework for why these pathways matter, this overview of metabolic health and longevity gives useful context on the larger physiological terrain.
What changes downstream of that signal
Once AMPK activation is the starting point, the downstream effects become easier to organize.
One expected outcome is greater glucose uptake into skeletal muscle, partly through mechanisms linked to GLUT4 translocation. A useful way to frame this is transport logistics. Glucose in the bloodstream is only potential fuel. GLUT4 helps move that fuel across the membrane and into the cell, where it can be used. In insulin-resistant states, that transport step is often impaired, so even a modest improvement can be mechanistically meaningful.
A second outcome is a shift toward fatty acid oxidation. Cells under energetic pressure often favor burning available substrate over storing it. MOTS-c appears to bias signaling in that direction in preclinical models, which helps explain why it continues to attract attention in studies of obesity, insulin resistance, and metabolic syndrome.
A third outcome is improved mitochondrial respiratory performance under stress conditions, along with lower reactive oxygen species emission in some model systems. That point needs careful wording. Lower ROS does not mean ROS are eliminated, and ROS are not purely harmful. They also function as signaling molecules. The more defensible interpretation is that MOTS-c may reduce damaging oxidative spillover while preserving the metabolic signaling network cells still need.
The mechanistic sequence can be summarized this way:
- MOTS-c triggers an energy-stress signaling response
- AMPK and PGC-1α pathways are required for key downstream effects
- Glucose transport and substrate oxidation increase
- Fat use rises when metabolic demand is high
- Mitochondrial respiration becomes more efficient in preclinical settings
- Oxidative stress markers fall in some experimental models
Mechanistic takeaway: The most credible MOTS-c hypothesis is improved control over cellular energy allocation, substrate use, and stress adaptation. That is a strong preclinical concept. It is not yet the same thing as proven therapeutic benefit in humans.
Primary MOTS-c Peptide Benefits in Metabolic Research
In preclinical metabolism studies, researchers pay attention when one intervention changes several linked endpoints at once. MOTS-c drew interest for that reason. In animal and cell models, it has been associated with lower weight gain under metabolic stress, better glucose handling, shifts in fuel selection, and less lipid accumulation in the liver. That combination is more informative than any single outcome on its own.
What the obesity and glucose data suggest
The clearest metabolic signal comes from high-fat diet models, where MOTS-c has been reported to blunt weight gain and improve glucose tolerance. As noted earlier, the original mouse work described meaningful effects on both adiposity-related outcomes and insulin action. For researchers, that pairing matters because body weight can change for many reasons, while glucose handling points more directly to altered metabolic control.
A useful analogy is traffic management in a crowded city. Weight gain is the traffic jam everyone notices. Glucose uptake, insulin sensitivity, and substrate use are the signaling system underneath. If those signals improve together, investigators have a stronger reason to suspect that the intervention is acting on coordination of energy flow rather than producing a superficial change in one readout.
That is also where caution is needed.
Most of these findings come from preclinical systems with controlled diets, defined dosing schedules, and relatively short study windows. Those conditions are valuable for mechanism work, but they do not automatically predict what will happen in heterogeneous human populations. Questions about dose, tissue exposure, treatment duration, and long-term safety still stand between an interesting mouse signal and a clinically useful therapy.
Researchers have also reported improved skeletal muscle glucose uptake, greater fatty acid use, reduced hepatic lipid buildup, and higher energy expenditure in these models. Taken together, those observations support a coherent hypothesis. MOTS-c appears to influence how the body allocates and uses fuel during metabolic stress, especially in muscle and liver.
These findings position MOTS-c as a regulator of metabolic stress responses, rather than solely a fat-loss agent.
Summary of preclinical metabolic outcomes
The table below organizes the main outcomes that keep MOTS-c relevant in metabolic research.
| Metabolic Parameter | Observed Effect in Preclinical Models | Significance for Research |
|---|---|---|
| Body weight gain | Reduced body weight gain under high-fat diet conditions | Useful for obesity and nutrient-overload models |
| Glucose tolerance | Lower blood glucose area under the curve during testing | Relevant for insulin resistance research |
| Insulin sensitivity | Improved handling of glucose in metabolically stressed animals | Supports work on metabolic syndrome pathways |
| Muscle glucose uptake | Greater uptake linked to GLUT4-related activity | Helpful in skeletal muscle metabolism studies |
| Fat oxidation | Shift toward using fatty acids as fuel | Relevant for substrate utilization questions |
| Hepatic lipid burden | Lower liver triglyceride accumulation in preclinical work | Useful in fatty liver and metabolic stress models |
| Energy expenditure | Increased heat output in animal studies | Supports whole-body metabolism investigations |
A few practical conclusions follow.
- For obesity models: MOTS-c is most informative when the study includes nutrient excess or clear metabolic strain.
- For insulin resistance research: glucose tolerance, insulin response, and muscle uptake endpoints are more informative than body weight alone.
- For mitochondria-centered experiments: pair MOTS-c with respiratory, redox, and substrate flux measurements to see the biology more clearly.
A scale can tell you that mass changed. It cannot tell you why. For MOTS-c, the more informative question is whether the intervention improved metabolic control under stress, and whether that effect can be reproduced across models with methods that are rigorous enough to support eventual translation.
Exploring Broader Applications in Longevity and Cellular Health
Aging research is full of compounds that look interesting in cell culture and then fade under the weight of physiology. MOTS-c has avoided that fate so far, at least in preclinical work, because some studies report system-level effects in older animals rather than isolated molecular changes. That is a higher bar. It still is not a clinical outcome.

Aging muscle and endurance capacity
One reason MOTS-c appears in longevity discussions is a finding from aged mice. In studies involving 22-month-old animals, treatment was associated with about a 2-fold increase in treadmill running time and distance, as summarized in this review-oriented overview of the literature, which also discusses inflammation findings in obese mouse models (Innerbody summary of preclinical findings).
That kind of result matters because endurance in older organisms is a composite trait. It reflects mitochondrial ATP production, substrate switching, cardiovascular support, neuromuscular coordination, and stress recovery. A change in treadmill performance is therefore less like a single blood marker and more like a systems stress test.
This is why MOTS-c attracts attention from researchers studying sarcopenia and functional aging.
Older muscle often shows the same broad failure pattern across different models. Mitochondria become less adaptable, redox control becomes less stable, and the tissue shifts less smoothly between fuels. MOTS-c is interesting in that context because its proposed biology sits upstream of those processes. It may influence how cells respond to energetic stress rather than pushing one output harder.
The exercise question is also important and still unresolved. Human studies suggest endogenous MOTS-c can rise with exercise, and some reports describe nuclear translocation in exercised muscle. That makes MOTS-c less like a replacement for training and more like a candidate signal within the adaptation network itself. Whether exogenous MOTS-c amplifies exercise responses, overlaps with them, or only matters under specific stress conditions remains an open experimental question.
Cardiac and inflammatory relevance
A second area of interest is tissue protection during metabolic strain, especially in organs with high energy demand. The heart is an obvious example because small disturbances in mitochondrial function can translate into measurable changes in contractile performance.
In diabetic rat cardiomyocytes and related animal work, MOTS-c was associated with restored mitochondrial oxidative phosphorylation capacity, reduced anoxic ATP hydrolysis, improved fasting glucose, and better echocardiographic function in that model (Frontiers in Physiology preclinical cardiac study). Researchers should read that carefully. These are preclinical data in a disease model, not evidence of a cardiac treatment effect in humans.
Inflammation adds another layer. Chronic low-grade inflammation often accompanies insulin resistance, vascular dysfunction, and declining physical performance in aging. In high-fat diet mouse studies summarized in the review above, chronic MOTS-c treatment was associated with lower TNF-α and IL-6. That does not prove MOTS-c is an anti-inflammatory therapy. It does suggest that changes in mitochondrial stress signaling may ripple outward into inflammatory tone, which is exactly the kind of cross-system effect aging biologists care about.
A useful analogy is wiring in a building. If the wiring is unstable, lights flicker in many rooms at once. You can chase each room separately, or you can inspect the electrical system. MOTS-c is being studied as part of that upstream wiring for cellular energy and stress adaptation.
For readers who want a quick visual explainer before returning to the primary papers, this overview is a reasonable starting point:
The cautious conclusion is straightforward. MOTS-c is a promising research tool for studying endurance, inflammatory tone, and tissue resilience in aging-related models. The barrier to translation is just as important as the promise. Dose, delivery, pharmacokinetics, reproducibility across labs, and relevance to human aging remain unresolved. Researchers sourcing material for that kind of work may compare documentation and specifications from suppliers such as Mots C.
Key Considerations for Researchers Studying MOTS-c
The easiest mistake in peptide research is to become more excited by the concept than by the material. With MOTS-c, that risk is high because the biology is appealing and the translational story is unfinished.
Why material quality matters
Experimental interpretation starts with material identity and consistency. If the peptide is impure, poorly documented, or inconsistently stored, you can’t confidently attribute an observed effect to MOTS-c itself.
For that reason, researchers should insist on basics that aren’t glamorous but are essential:
- Certificate review: verify the lot has a COA and matching batch documentation.
- Analytical traceability: look for microbial and endotoxin reporting when relevant to the model.
- Handling discipline: follow reconstitution, storage, and freeze-thaw limits appropriate to the material.
- Protocol fit: choose endpoints that match the hypothesized mechanism, not just broad wellness claims.
If you’re comparing vendors or building a procurement checklist, a product listing such as Mots C from Peptide warehouse Shop can serve as one example of how suppliers present research-use MOTS-c information for evaluation.
This is also the one place where Peptide Warehouse USA fits naturally into the discussion. The company provides research peptides with batch documentation, COAs, and related testing reports for laboratory and analytical use, which is the sort of documentation framework researchers should look for before starting a study.
Why preclinical promise has not yet become clinical routine
Careful interpretation is paramount. Despite promising animal data, effective clinical translation has been lacking, and MOTS-c is not known to be blood-brain barrier penetrant, which limits claims about direct cognitive application in human studies.
That single point clears up a lot of confusion. A peptide can show neuroprotective or cognition-related signals in preclinical systems without being ready for direct human cognitive use. Delivery, stability, tissue penetration, formulation, and regulatory development all matter.
Practical rule: treat MOTS-c as a research compound with promising biology, not as a clinically resolved intervention.
Practical design questions for lab teams
If you’re designing a study, three questions are more useful than broad enthusiasm.
First, what tissue is your primary target? Skeletal muscle, liver, adipose tissue, and cardiac tissue may each require different readouts.
Second, is your model metabolically stressed? Much of the strongest MOTS-c signal appears in high-fat diet, diabetic, aged, or otherwise challenged systems. A healthy unstressed model may show less dramatic separation.
Third, are you measuring mechanism or only phenotype? Body weight and treadmill time are useful, but mitochondrial respiration, ROS output, GLUT4-related endpoints, and AMPK-pathway markers often provide the stronger interpretive value.
A reasonable research workflow often includes:
- selecting a metabolically relevant model,
- validating peptide quality,
- pairing phenotype endpoints with mechanistic assays,
- interpreting results with clear preclinical limits.
That approach won’t make the biology simpler, but it will make your conclusions stronger.
Conclusion The Future of MOTS-c in Research
The most credible case for MOTS-c peptide benefits is also the most disciplined one. MOTS-c is a mitochondrial-derived peptide with meaningful preclinical evidence in metabolic regulation, glucose handling, mitochondrial bioenergetics, endurance, inflammation, and cardiac stress models. Mechanistically, the strongest story centers on AMPK and PGC-1α-dependent effects rather than vague claims about “cellular energy.”
The limitations are just as important as the benefits. Human translation remains incomplete, and current evidence doesn’t justify treating MOTS-c as a settled clinical tool. For researchers, that isn’t disappointing. It’s exactly what makes the compound worth studying carefully.
If you’re tracking body composition or performance adaptations in a broader training context, tools that clarify phenotype can help frame your experimental thinking. Resources like DEXA scan insights for athletes are useful for understanding how researchers and practitioners think about body composition beyond scale weight.
The field still needs better translational work, better delivery strategies, and clearer human data. Until then, MOTS-c belongs in the category of promising, mechanistically interesting, research-first compounds. Learn more, explore options carefully, and treat the evidence with the precision it deserves.
If you’re sourcing MOTS-c or related compounds for laboratory, analytical, or preclinical work, Peptide Warehouse USA offers a research-use catalog with batch-based documentation, including COAs and related testing records. For teams that value traceability and US-based supply, it’s a practical place to explore options and review available peptide materials.






Leave a comment