Expert Guide: what is bpc 157 Explained
Inquiries about what is BPC-157 often treat the answer as a mere label. It isn’t. For a lab manager or serious peptide buyer, the pertinent question is more specific: what exactly is this compound, what has it done in preclinical models, and where do the scientific and regulatory boundaries sit?
That gap matters. BPC-157 gets discussed online like a finished therapy, but the evidence base is still heavily preclinical and the regulatory status is clear. It’s an unapproved research peptide, not an FDA-approved drug, and that distinction should guide how you evaluate data, sourcing, and intended use.
Table of Contents
- The Molecular Identity and Origin of BPC-157
- Proposed Mechanisms of Action in Research Models
- A Summary of Preclinical Research Evidence
- Common Research Stacks Involving BPC-157
- Understanding BPC-157 Safety and Regulatory Status
- Sourcing High-Purity BPC-157 for Laboratory Use
- Frequently Asked Questions About BPC-157 Research
The Molecular Identity and Origin of BPC-157

What BPC-157 is at the chemical level
BPC-157 is a synthetic peptide made of 15 amino acids. Its sequence is Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val, and it was derived from a protective protein associated with human gastric juice, as described in the background summary on BPC-157.
That definition clears up a common confusion. BPC-157 is not a plant extract, not a vitamin, and not a broad “supplement blend.” It’s a discrete, sequence-defined research compound. For labs, that matters because sequence-defined compounds can be evaluated for purity, identity, and batch consistency in a way that herbal or mixed products often can’t.
Another point often missed is the name itself. You may see it referred to as Body Protection Compound 157 or gastric pentadecapeptide BPC-157. In practical research terms, both names refer to the same peptide identity.
Practical rule: If a vendor can’t clearly state the peptide identity and sequence, you can’t treat the material as a serious analytical or preclinical input.
Why its origin and stability matter
BPC-157 first drew interest during research conducted by Croatian scientists in the early 1990s. That origin story isn’t just historical trivia. It explains why the peptide became associated with gastric protection and tissue repair in the first place.
Its stability is one of the most notable features. The peptide has been described as remaining intact in human gastric juice for over 24 hours in the same background source linked above. For researchers, that’s not a marketing detail. Stability affects handling assumptions, formulation questions, and how a compound is expected to behave in harsh biological environments.
This also explains why BPC-157 has held attention for so long in regeneration research. A peptide that remains stable under acidic conditions stands out immediately because many peptides degrade quickly in less challenging settings.
A simple way to think about it is this:
- Identity tells you what it is.
- Origin tells you why researchers noticed it.
- Stability tells you why they kept studying it.
That combination is why the question “what is bpc 157” deserves more than a one-line answer. It’s a chemically defined peptide with a specific research history, not a vague wellness trend.
Proposed Mechanisms of Action in Research Models
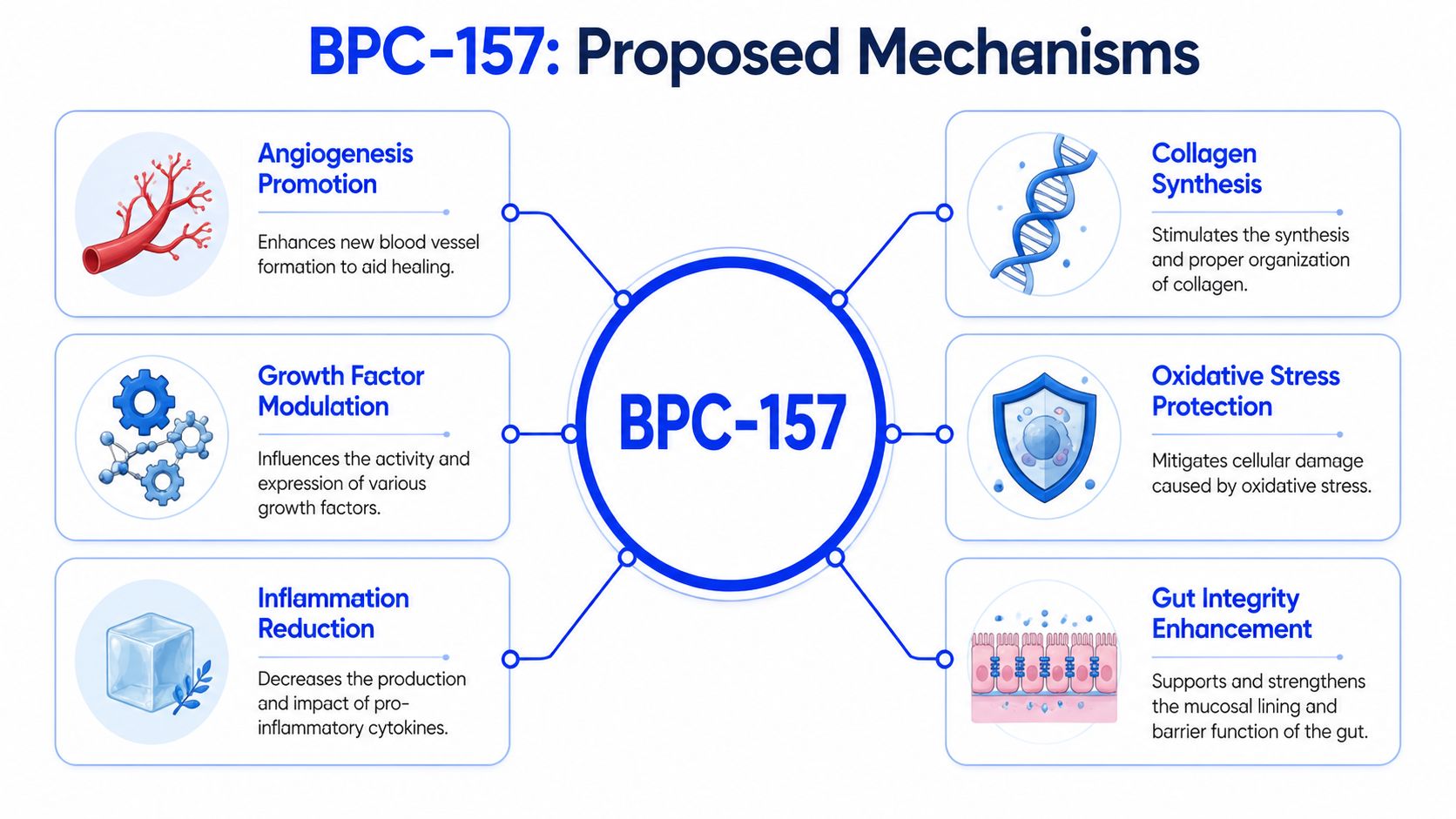
FAK-paxillin and fibroblast signaling
Researchers don’t study BPC-157 just because it exists. They study it because preclinical work suggests it may influence pathways tied to repair. One proposed mechanism involves the FAK-paxillin pathway, along with upregulation of Growth Hormone Receptors on fibroblasts, as described in this mechanistic overview.
In plain language, fibroblasts are among the workhorse cells of connective tissue repair. They help build and organize extracellular matrix, including collagen. If a compound alters fibroblast behavior, researchers pay attention because tendon, ligament, and wound healing depend heavily on those cells.
FAK and paxillin are part of the signaling machinery that helps cells attach, move, and respond to their surroundings. A useful analogy is scaffolding and navigation. Cells repairing damaged tissue need both a place to anchor and instructions on where to move. Pathways like FAK-paxillin help coordinate that behavior.
Angiogenesis and repair signaling
Another recurring theme in BPC-157 research is angiogenesis, or the formation of new blood vessels. That’s important because injured tissue doesn’t just need structural repair. It needs blood flow, oxygen delivery, and nutrient support.
When readers get confused here, it’s usually because “healing” sounds like one process. It isn’t. In research models, repair often depends on several linked processes happening together:
- Cell migration: repair cells must reach the damaged site.
- Collagen organization: tissue has to be rebuilt, not just filled in.
- Vascular support: new or improved blood supply helps sustain the repair environment.
- Signal amplification: receptor and pathway changes can make repair cues more effective.
BPC-157 is best understood as a compound of mechanistic interest, not a settled clinical solution.
Some preclinical discussions also connect BPC-157 to VEGF-related signaling and endothelial activity. That’s one reason it appears so often in conversations about connective tissue and poorly vascularized structures. Tendons and ligaments are notoriously slow to heal, partly because blood supply is limited compared with other tissues.
For a research scientist, the key takeaway isn’t “this peptide heals everything.” The takeaway is narrower and more useful: BPC-157 is a candidate compound for studying how peptide signaling may influence fibroblast activity, tissue organization, and vascular support in injury models.
A Summary of Preclinical Research Evidence
What tendon models have shown
The strongest interest around BPC-157 comes from animal work, especially musculoskeletal models. One widely cited example comes from rat Achilles tendon transection research, where BPC-157 at 10 μg/kg significantly accelerated functional recovery, and treated groups showed higher load-to-failure values and superior tissue remodeling over a 14-day period, according to this published preclinical report.
That kind of result matters because tendon repair can be measured in more than one way. Researchers don’t only ask whether tissue looks better under microscopy. They also ask whether the repaired structure functions better mechanically. In tendon studies, those are not the same thing.
BPC-157 particularly stands out in the preclinical conversation. It has been examined not only for visible remodeling but also for functional outcomes in injury models.
Key areas of preclinical interest
The broader animal literature around BPC-157 spans several categories. The findings are heterogeneous, but the research themes are fairly consistent.
| Research Area | Observed Outcome in Animal Models | Common Model Used |
|---|---|---|
| Tendon healing | Improved functional recovery, stronger repair, more organized tissue remodeling | Achilles tendon transection in rats |
| Muscle and soft tissue repair | Faster recovery signals and improved structural healing are commonly investigated | Traumatic injury models in rodents |
| Gut and cytoprotection | Protective effects in gastrointestinal injury contexts are a long-running area of interest | Gastric and intestinal injury models |
| Vascular response | Endothelial and blood flow related effects are studied in ischemic and repair settings | Rodent ischemia and injury models |
A careful reader should notice what’s not in that table. There are no large human randomized trials listed, because that isn’t where the evidence is strongest. The center of gravity remains preclinical.
The animal data are intriguing because they connect mechanism to function. The same fact also sets the limit. Animal success does not equal clinical validation.
If you’re evaluating what is bpc 157 from a scientific perspective, that’s the correct frame. It’s a peptide with sustained preclinical interest, especially in tendon and tissue-repair models, but the evidence base still sits far closer to the bench than the clinic.
Common Research Stacks Involving BPC-157

Why researchers combine compounds
In peptide research, a “stack” usually means studying more than one compound in the same protocol to examine complementary effects. That approach isn’t unique to BPC-157. It reflects a basic experimental idea: tissue repair is multi-factorial, so some investigators design studies around compounds that may act through different pathways or tissue distributions.
BPC-157 often appears in these conversations because its preclinical reputation centers on connective tissue and repair signaling. Researchers may pair it with another peptide to ask a more specific question, such as whether local tissue organization and broader recovery signaling can be studied together within one model.
A sound stack hypothesis should answer three questions:
- Why these compounds together
- What distinct mechanism each one is supposed to contribute
- How the protocol will separate overlap from actual synergy
Without that discipline, stacking turns into guesswork.
BPC-157 and TB-500 in study design
The most common comparison is BPC-157 versus TB-500, or BPC-157 used alongside TB-500 in research discussions. The practical distinction researchers often make is conceptual. BPC-157 is commonly discussed in relation to localized healing processes, while TB-500 is often framed as a broader systemic recovery peptide in experimental conversations.
That doesn’t mean a stack is automatically smarter than a single-compound design. In fact, stacking can make interpretation harder if endpoints aren’t well chosen. If both compounds influence overlapping recovery pathways, the lab may struggle to determine which input drove which result.
A useful way to approach stack design is to define separate readouts:
- Histology-focused endpoints for tissue architecture
- Biomechanical endpoints for strength and function
- Timing comparisons to see whether one compound appears to affect early versus later repair phases
For readers who want a quick visual walkthrough of common peptide stacking discussions, this overview is a useful supplement:
In other words, stacks belong in carefully controlled research design, not in casual assumptions. If your endpoint is murky, adding another peptide won’t fix it.
Understanding BPC-157 Safety and Regulatory Status
Why unapproved does not mean useless
A compound can be scientifically interesting and still remain unapproved. That’s the category BPC-157 falls into. As of 2022, BPC-157 was banned by the World Anti-Doping Agency under the S0 category of non-exempt substances, and it lacks approval from major drug regulatory agencies like the FDA for human use, according to the background summary cited in the earlier source section.
That combination creates a lot of confusion online. Some readers assume “not approved” means “no science.” Others assume “widely discussed” means “clinically accepted.” Neither is accurate.
Unapproved means the compound has not cleared the regulatory standard required for approved human medical use. It does not erase the preclinical literature. It does mean labs, buyers, and readers should keep the compound in its proper category: research only.
Why banned in sport is not the same as approved in medicine
The WADA issue deserves its own clarification because people often misread it. A sports ban and a medical approval are different systems with different goals.
WADA evaluates whether substances should be prohibited in competition. Drug agencies evaluate whether a substance has sufficient evidence for safety, quality, and efficacy in approved medical use. A ban in sport doesn’t convert a peptide into an approved therapy, and lack of approval doesn’t automatically explain why sports regulators prohibited it.
That distinction matters for purchasing language, internal lab policy, and public-facing education.
- For labs: keep labeling and intended-use language strictly non-clinical.
- For buyers: don’t interpret athlete interest as proof of validated human benefit.
- For compliance teams: separate sports-policy language from drug-approval language.
Research enthusiasm should never blur category boundaries. BPC-157 belongs in the investigational bucket, not the approved-therapy bucket.
The safety discussion is also incomplete without acknowledging uncertainty. Human data remain limited, so quality control and documentation become even more important. When evidence is early, material quality matters more, not less.
Sourcing High-Purity BPC-157 for Laboratory Use
What to verify before you buy
How much confidence can a lab place in a BPC-157 experiment if the material in the vial is only loosely documented?
For researchers, sourcing is part of study design. A peptide can look fine on a product page and still introduce uncertainty at the bench through impurity, degradation, mislabeling, or poor lot control. In practice, the purchasing decision works like the foundation under an assay. If the foundation is weak, every downstream observation becomes harder to interpret.
Start with records that tie directly to the lot in hand. A supplier should be able to show what was tested, which batch was tested, and how that batch connects to the item shipped to your lab.
A useful review checklist includes:
- Batch-specific COA: the certificate should correspond to the exact lot purchased, not a generic reference file.
- Identity and purity data: the documentation should state what analytical method was used and what purity result was reported for that lot.
- Microbial and endotoxin reporting: these data are often relevant for preclinical and analytical workflows where contamination can distort results.
- Lot traceability: labels, invoices, and test records should align clearly enough for internal audit or protocol review.
- Storage and handling guidance: peptide stability depends in part on how material is shipped, stored, and reconstituted in the lab.
Why documentation affects experimental confidence
In an early-stage research setting, quality paperwork is not administrative clutter. It is part of the evidence chain.
If one batch is less pure than another, your readout may reflect batch variation rather than compound biology. If the peptide has degraded during storage, a failed replication attempt may say more about handling history than mechanism. This is the gap many research buyers underestimate. Exciting preclinical findings create interest, but reproducible lab work still depends on ordinary procurement discipline.
That is why the logic behind understanding independent supplement testing carries over surprisingly well. The article discusses supplements, but the central principle also applies to research peptides. Independent verification gives a lab a better starting point than supplier claims alone.
Peptide Warehouse USA is one example of a supplier that describes research-product documentation such as COAs, microbial reports, endotoxin reports, and stated purity values. The specific vendor matters less than the standard being applied. A serious buyer should ask whether the documentation is batch-linked, readable, and sufficient for internal quality review.
For BPC-157, that standard matters more because the compound sits in an unusual category. Preclinical interest is real. Regulatory approval for human medical use is absent. In that kind of research-only market, careful sourcing is not a marketing preference. It is one of the few controls a lab can apply before the experiment begins.
Frequently Asked Questions About BPC-157 Research
Is BPC-157 legal to buy for research
In many discussions, the more precise issue isn’t simple legality. It’s intended use. BPC-157 is commonly sold in the research market as a research chemical or investigational compound, and labs should keep purchasing, labeling, and internal handling aligned with non-human research use.
Is BPC-157 approved for human use
No. It is not approved by major agencies such as the FDA for human use. That’s why any serious educational material should avoid treating it like an established medical therapy.
What should a lab prioritize when evaluating a supplier
Start with documentation and consistency. A supplier should be able to provide batch-linked testing records, clear product identity, and handling information that supports analytical or preclinical use.
Then look at process quality:
- Lot transparency: can you trace one batch from order to documentation?
- Testing depth: are purity and contamination controls both addressed?
- Research alignment: does the company clearly state research-only use?
- Operational clarity: are shipping, storage, and support details easy to verify?
A final point often gets overlooked. If your team can’t explain why it chose one supplier over another in a single page of internal documentation, the selection process probably wasn’t rigorous enough.
BPC-157 remains compelling because the preclinical signal is hard to ignore, especially in tissue-repair research. But the responsible stance is straightforward. Treat it as a promising, unapproved peptide of laboratory interest, and hold sourcing standards high enough that your data mean something.
If you’re evaluating research-grade peptide options, Peptide Warehouse USA offers BPC-157 and related compounds for laboratory, analytical, and preclinical use, with batch documentation designed to support traceability. Learn more and explore options if your team needs a research-only supplier with clear COA and testing records.






Leave a comment