Is Bacteriostatic Water the Same as Reconstitution Solution?
No, bacteriostatic water is not the same as reconstitution solution. Bacteriostatic water is a specific reconstitution solution made with sterile water plus 0.9% benzyl alcohol, while reconstitution solution is a broader category that can include preservative-free diluents intended for single use.
That distinction looks minor until a lab starts losing consistency across runs, throwing out partially used vials, or introducing avoidable contamination risk during repeat withdrawals. In peptide work especially, the diluent isn’t just a helper liquid. It affects how long a vial remains usable, how often it can be accessed, how stable the material remains in storage, and whether your handling process supports clean, repeatable results.
A lot of confusion comes from the language people use on forums, vendor listings, and even inside labs. “BAC water,” “sterile water,” and “reconstitution solution” often get treated as if they’re interchangeable. They aren’t. If you’re training new staff, setting SOPs, or tightening budget controls around peptide handling, this is one of those basic decisions that has real downstream consequences.
Table of Contents
- Introduction The Critical Distinction in Your Lab
- Defining the Fundamentals Reconstitution Solution and Bacteriostatic Water
- A Head-to-Head Comparison Key Differences That Matter
- The Case for Bacteriostatic Water in Peptide Research
- When to Choose Alternative Reconstitution Solutions
- A Practical Guide to Reconstituting Research Peptides
- Frequently Asked Questions for Researchers
- What should go into the lab record when a peptide is reconstituted
- When is aliquoting a better choice than keeping one working vial
- What is the most common documentation mistake after reconstitution
- How do I decide whether a preservative is acceptable in my workflow
- When should a reconstituted vial be discarded even if material remains
- Can one lab standardize on a single diluent for all peptides
Introduction The Critical Distinction in Your Lab
If you’re asking whether is bacteriostatic water the same as reconstitution solution, the useful answer is this: one is a category, the other is a standardized member of that category. Treating them as identical can weaken sterility control, distort storage expectations, and create avoidable waste.
In a research setting, diluent choice affects more than convenience. It shapes how often you can return to the same vial, how long a reconstituted material remains workable, and whether your process supports reproducibility across technicians and study days.
A new team member usually sees only one step: add liquid to powder. A lab manager sees the chain behind that step. Which vial can be reopened. Which peptide lot can stay in rotation. Which assay might be sensitive to preservatives. Which SOP is easier to audit when questions come up later.
Practical rule: If a protocol involves repeated withdrawals over time, the diluent decision becomes a workflow decision, not just a chemistry decision.
That’s why this topic matters. The right choice protects experiment integrity and supports cost control. The wrong choice can leave you with unstable material, excess discard volume, or a preventable compliance headache.
Defining the Fundamentals Reconstitution Solution and Bacteriostatic Water
Reconstitution solution is a job description
Reconstitution solution isn’t one fixed product. It’s a functional term for any sterile diluent used to dissolve a lyophilized compound so that the material can be handled, measured, or studied in solution.
That means several liquids can fit under the label depending on the protocol. A preservative-free sterile water product may count. So can saline in some workflows. The point is function, not a single universal formula.
Consider it the “vehicle” in a lab notebook. This term describes the liquid’s specific function. It does not provide a precise list of the bottle’s ingredients.

Problems arise when teams purchase supplies without verifying their composition. A label reading “Reconstitution solution” might appear appropriate, yet you must still confirm if it includes a preservative, if it is designed for multi-use handling, and if it is compatible with the specific compound and assay.
For labs that care about upstream water quality and filtration processes in general manufacturing or prep environments, this overview of choosing nano filtration membranes is a useful background read. It helps newer staff understand why not all purified water systems produce water with the same downstream handling implications.
Bacteriostatic water is a defined product
Bacteriostatic water is not just any diluent. It’s a specifically defined sterile water formulation containing 0.9% benzyl alcohol as a preservative, as described in this review of reconstitution solution vs bacteriostatic water.
That preservative matters because it gives the product a bacteriostatic effect. It inhibits bacterial growth rather than actively killing bacteria. Operationally, that shift is what allowed labs and clinical settings to move away from purely single-use sterile water workflows toward multi-dose vial systems.
The hierarchy becomes clear:
- Reconstitution solution: the broad category
- Bacteriostatic water: one specific product inside that category
- Sterile preservative-free options: other products that may also fall inside the category
All bacteriostatic water is a reconstitution solution. Not all reconstitution solutions are bacteriostatic water.
That distinction sounds simple, but it clears up most of the confusion. If your team documents “reconstitution solution” without naming the actual formulation, your records may be technically correct yet operationally incomplete. For sensitive compounds and repeated handling, that missing detail matters.
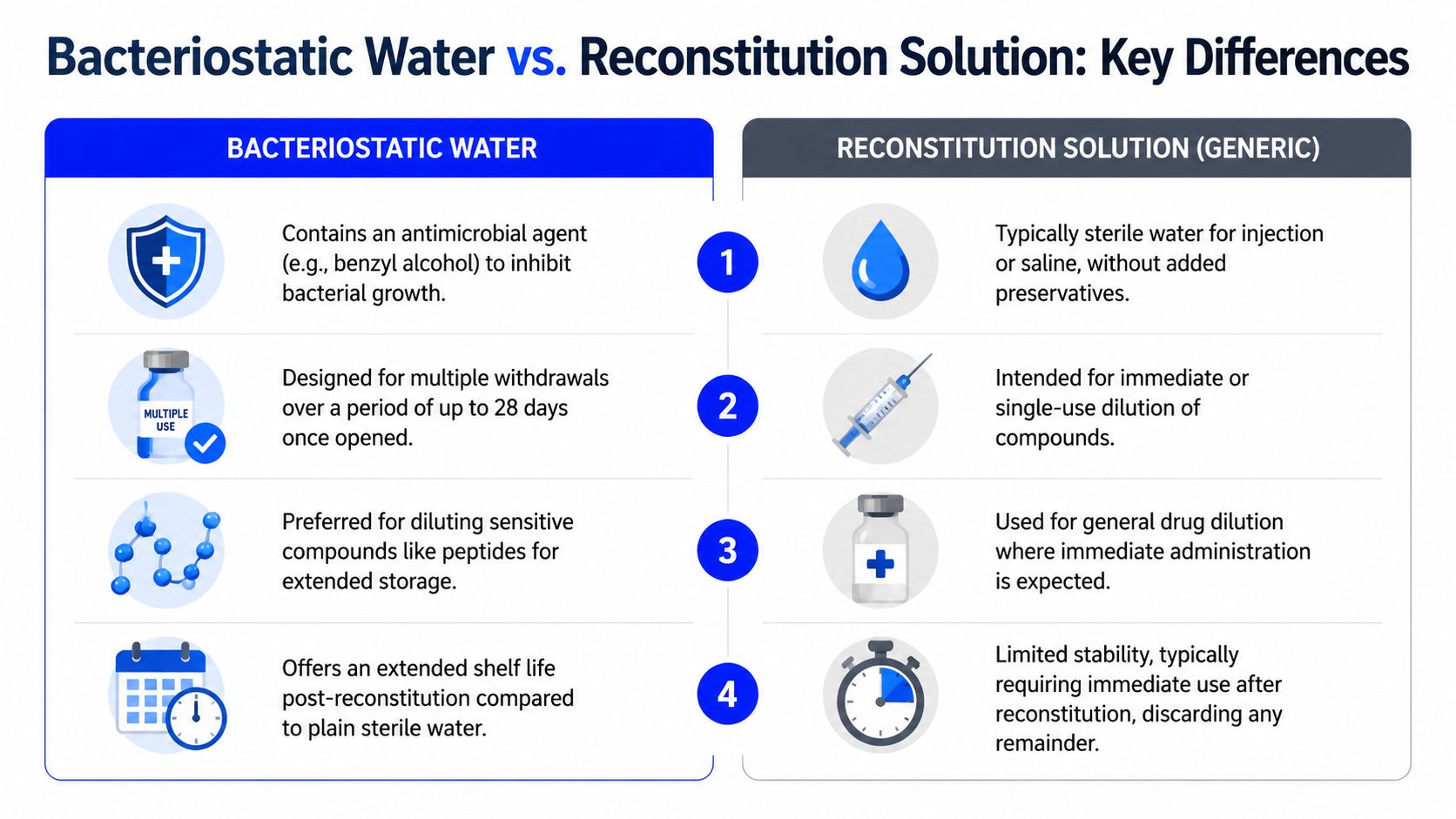
A Head-to-Head Comparison Key Differences That Matter
Which choice protects your sample, your budget, and your records once the vial is no longer factory-sealed? In routine lab work, that is the comparison that matters.
Comparison table for daily lab decisions
Teams run into trouble when they treat a broad label and a defined formulation as if they carry the same handling rules. They do not. One gives you a specific preservative-containing product with a known reuse model. The other only tells you the liquid is being used to reconstitute something.
| Feature | Bacteriostatic Water | Generic Reconstitution Solution |
|---|---|---|
| What it is | A specific sterile water formulation with 0.9% benzyl alcohol | A broad category for sterile diluents used to dissolve compounds |
| Preservative | Yes | May or may not contain one |
| Typical use pattern | Multi-dose handling | Often single-use handling |
| Opened vial use window | Commonly labeled for repeated use after opening when handled aseptically, subject to product instructions and institutional policy | Preservative-free options are usually treated as single-use or limited-use materials |
| pH profile | Controlled by the product specification | Depends on the specific formulation |
| Best fit | Repeated withdrawals, longer handling windows, workflows that benefit from a defined preservative system | Immediate prep, single-session work, preservative-sensitive applications |
What changes once the vial is opened
The operational split shows up after first puncture. On the shelf, two sterile diluents may look interchangeable. At the bench, they create different risk profiles for contamination control, documentation, and waste.
Bacteriostatic water supports repeated access because the formulation includes a preservative. A generic reconstitution solution may be sterile and still be a poor fit for repeat entry if it is preservative-free. That difference affects scheduling. It also affects cost. If staff must discard partially used diluent after each prep session, supply use climbs and variability between prep events climbs with it.
In practice, the wrong choice usually creates one of three problems:
- Higher discard volume: partially used vials leave service sooner
- More process variation: each fresh prep introduces another chance for concentration or handling drift
- More documentation burden: staff must log more vial openings, lot numbers, and preparation events
I usually tell new staff to choose the handling model first, then choose the diluent. If the protocol involves repeated withdrawals, the diluent has to support that pattern without forcing workarounds.
Why formulation control matters
A named product gives a lab something a generic category cannot. It gives a fixed specification that can be written into SOPs, training records, deviation reviews, and purchasing controls.
That matters for experiment integrity. If one technician uses bacteriostatic water, another uses preservative-free sterile water, and both write “reconstitution solution” in the notebook, the record hides a meaningful variable. If results shift later, root-cause review gets harder because the diluent was never documented precisely.
It matters for cost control too. Standardizing one defined diluent for the workflows that require repeated vial access reduces ad hoc substitutions, unnecessary disposals, and purchasing confusion. Standardizing the wrong one creates the opposite problem. A preservative-containing product used where the compound is preservative-sensitive can compromise the material. A preservative-free product used for repeated access can increase waste and handling risk.
The practical rule is simple. Document the exact diluent, not the category name, and match it to the access pattern your protocol requires.
The Case for Bacteriostatic Water in Peptide Research
Peptide workflows expose the practical value of bacteriostatic water very quickly. These studies often involve small vials, repeated withdrawals, and strong incentives to preserve sample quality over time. In that setting, the preservative isn’t just a convenience feature. It supports the entire handling model.
Where it earns its place
In peptide research, bacteriostatic water’s 0.9% benzyl alcohol content inhibits more than 99% of bacterial growth, supports 20-30 punctures with less than 0.1% contamination risk, and extends peptide potency retention by 15-20% over 14 days compared with sterile water, based on this analysis of bacteriostatic water for peptide reconstitution.
Those numbers line up with what experienced labs already notice in routine handling. When a peptide vial must stay useful across a schedule instead of a single prep session, bacteriostatic water fits the workflow better than a generic single-use diluent.
This matters most when your protocol includes:
- Repeated sampling: You expect to return to the same reconstituted vial multiple times
- Small-dose workflows: The lot value sits in many withdrawals, not one large use
- Longer study windows: The handling period extends beyond a single bench session
Operational benefits you notice quickly
The operational benefits are easy to spot before you ever discuss chemistry in depth.
First, staff make fewer replacement vials. Second, fewer partially used diluent containers get discarded. Third, the SOP becomes clearer because the same product is used for the same class of work every time.
That consistency protects your study in quiet ways. Fewer ad hoc substitutions. Fewer labeling ambiguities. Fewer moments where one technician assumes “reconstitution solution” means the same thing another technician used last week.
Multi-dose peptide protocols work best when the diluent is chosen for repeated access from the start, not justified after the fact.
There’s also a budget point that many labs underestimate. Premium research materials lose value fast when the handling system around them is casual. A cheaper single-use diluent can become the expensive choice when it drives more waste, more remakes, and more discarded partially used stock.
When to Choose Alternative Reconstitution Solutions
Which diluent protects the study better: the one that extends vial usability, or the one that introduces the fewest variables into the assay?
The answer depends on where the risk sits in your workflow. In some protocols, bacteriostatic water improves handling control. In others, the preservative becomes an avoidable source of assay interference, documentation complexity, or failed repeats. The practical decision is not about preference. It is about protecting readouts, limiting waste, and staying aligned with the method.
Cell-based work needs extra caution
Cell culture is the clearest example. Benzyl alcohol helps control microbial growth in multi-use handling, but that same preservative can be a poor fit for sensitive cell systems. In assay development, I treat peptide solubility and biological compatibility as separate checks for exactly this reason.
A peptide may reconstitute cleanly in bacteriostatic water and still create downstream problems once that stock enters a cell-based assay. If the model is sensitive, even a small amount of preservative carryover can affect viability, morphology, or signal quality. That creates a bad trade. The lab gains convenience at the reconstitution step, then pays for it in noisy data or repeat work.
For cell-based protocols, review three things before selecting the diluent: the peptide’s known solubility requirements, the assay matrix, and the tolerated excipient profile in the final well or sample. If the method cannot tolerate benzyl alcohol, use a preservative-free sterile diluent and control contamination risk through single-use handling and tighter prep discipline.
Single-use workflows often don’t need a preservative
A preservative only earns its place when it solves a real handling problem.
If the vial will be reconstituted, used once, and discarded in the same session, a simpler sterile diluent often makes more operational sense. It removes one compatibility variable, simplifies review during deviation investigations, and may reduce unnecessary spend on specialty diluents for work that never involves repeat access.
Alternative reconstitution solutions are often the better choice in workflows such as:
- Immediate-use preparation: The full contents are used in one session
- Preservative-sensitive assays: The method, matrix, or detection system reacts poorly to added excipients
- Strict analytical applications: The protocol favors the cleanest possible background
- High-value samples with narrow acceptance criteria: Extra formulation components increase risk during method transfer or troubleshooting
There is also a compliance angle. If your SOP describes single-use preparation, but staff reach for bacteriostatic water out of habit, the lab creates preventable inconsistency between protocol intent and bench practice. That may not show up as a dramatic failure. It shows up later as harder root-cause analysis, uneven recordkeeping, and questionable comparability across runs.
Choose the diluent that fits the assay, not the one that feels broadly useful. Bacteriostatic water is a strong option for repeated withdrawals. Alternative reconstitution solutions are the better tool when assay sensitivity, single-use handling, or method restrictions make preservative exposure a liability.
A Practical Guide to Reconstituting Research Peptides
What ruins more peptide work in the lab: the powder, or the reconstitution step? In practice, reconstitution errors cause a large share of avoidable variability because they affect concentration accuracy, contamination risk, sample stability, and how confidently a result can be defended later.
For laboratory work, technique matters as much as diluent selection. A suitable solution cannot rescue poor handling, incomplete records, or inconsistent storage. These materials are for research, laboratory, or analytical use only, and they should be handled under controlled conditions with documented aseptic practice.

Before you puncture the vial
Preparation starts before the stopper is touched. Clear the bench, confirm the current SOP, and gather everything needed for one uninterrupted workflow: the peptide vial, the selected diluent, sterile syringes, transfer components if your procedure separates draw and transfer steps, alcohol swabs, labels, and the correct storage container.
Then verify four points before drawing any liquid:
- Confirm the diluent identity: Read the label. Do not rely on cap color, box placement, or memory.
- Confirm method compatibility: Check that the peptide and the downstream assay can tolerate the chosen diluent and any preservative it contains.
- Inspect container integrity: Reject vials with damaged stoppers, poor labeling, visible contamination, or uncertain storage history.
- Set the target concentration in advance: Decide the reconstitution volume first so the math, aliquot plan, and storage plan are all aligned.
This step protects both data quality and budget. A concentration error made here often leads to repeat prep, wasted peptide, and results that cannot be compared cleanly across runs.
How to add the diluent without damaging the sample
Disinfect vial stoppers and let them dry fully. Draw the planned volume with sterile technique, then add the diluent slowly against the inside wall of the peptide vial instead of forcing a stream directly onto the powder cake. That approach reduces foaming and helps fragile material wet evenly.
Do not shake aggressively.
Some peptides dissolve within seconds. Others need time, gentle swirling, or a controlled rest period. If a vial does not clear quickly, forcing dissolution can create a second problem by introducing shear, bubbles, or inconsistent recovery. Follow the handling notes for that specific material and record any deviation from the routine method.
A practical training point for new staff is simple: the goal is not just to get the powder into solution. The goal is to get a known concentration, with minimal stress on the sample, in a form that can be used again or discarded according to the protocol without ambiguity.
Aseptic technique affects more than cleanliness. It affects whether a multi-use vial remains usable, whether an out-of-spec result can be investigated, and whether the lab avoids preventable waste.
Storage labeling and routine handling
Once reconstituted, label the vial immediately with the compound name, diluent used, final concentration, date of reconstitution, and required storage condition. If your system tracks user initials, lot numbers, or expiry assignment, record those at the same time. Delayed labeling is a common source of mix-ups, especially in shared cold storage.
Bacteriostatic water can reduce contamination risk and unnecessary discard in workflows that involve repeated withdrawals, but those benefits depend on disciplined handling. Preservative or not, a poorly managed vial is still a weak point in the process.
A reliable routine usually includes:
- Date every vial: Undated material should not stay in active use
- Record the exact diluent: “Reconstitution solution” is too vague for deviation review or repeatability
- Minimize bench exposure: Return prepared material to proper storage promptly
- Check appearance before reuse: Cloudiness, particles, color change, or unexpected precipitation require review against the protocol
- Limit unnecessary punctures: Repeated access has a cost, even in multi-dose workflows, so aliquot when the use pattern supports it
Good reconstitution practice is operational control at the bench level. It protects sample integrity, keeps reruns down, supports SOP compliance, and makes better use of expensive peptide inventory.
Frequently Asked Questions for Researchers
What should go into the lab record when a peptide is reconstituted
Record the peptide name, lot number, diluent, final concentration, preparation date, preparer, and assigned storage condition. If the vial will be used across multiple runs, add the planned in-use window and any aliquot IDs.
That level of detail is not paperwork for its own sake. It is what lets a team trace a failed run back to a preparation variable instead of repeating the entire experiment with no clear corrective action.
When is aliquoting a better choice than keeping one working vial
Aliquot when the material is expensive, the study runs over multiple days, or the vial would otherwise be punctured repeatedly. A few extra tubes cost less than losing a peptide batch to contamination, adsorption, or freeze-thaw damage.
I usually treat aliquoting as a cost-control decision as much as a handling decision. It reduces avoidable waste and makes deviation review easier.
What is the most common documentation mistake after reconstitution
Writing “reconstitution solution” without naming the exact diluent. That wording is too broad for reproducibility, audit review, or cross-study comparison.
If one analyst used bacteriostatic water and another used sterile water or saline, the label needs to show that clearly. Otherwise, the lab creates a preventable source of variability.
How do I decide whether a preservative is acceptable in my workflow
Start with the full workflow, not just the reconstitution step. Review assay chemistry, cell exposure, injection volume, storage duration, and how many times the vial will be accessed. A preservative may help one workflow and interfere with another.
This decision should sit in the method or SOP, not in personal habit at the bench.
When should a reconstituted vial be discarded even if material remains
Discard it if labeling is incomplete, storage history is uncertain, appearance has changed, or the vial was handled outside the approved procedure. Retaining questionable material to save cost often creates a larger cost later through invalid data, reruns, and failed comparability.
Can one lab standardize on a single diluent for all peptides
That approach simplifies purchasing, but it is rarely the best technical choice. Standardizing too aggressively can create compatibility problems or force avoidable exceptions later. A better system is to standardize decision rules, approved diluents, and labeling requirements, then assign the diluent by compound and method.
If you’re sourcing high-purity research peptides and want a supplier built around traceability, batch documentation, and laboratory-focused handling standards, learn more about Peptide Warehouse USA. You can explore options for research peptides and related compounds backed by COAs, microbial and endotoxin reporting, and clear research-use-only positioning.






Leave a comment