Reconstituting AOD 9604: A Step-by-Step Research Guide
You’re probably here with a vial of lyophilized AOD 9604, bacteriostatic water on the bench, and one concern above all others. You don’t want to ruin the batch.
That concern is justified. Reconstituting AOD 9604 looks simple until the powder clumps, turns cloudy, or gels into something unusable. Most failures don’t come from bad intent. They come from rushed solvent addition, cold diluent, rough handling, or sloppy math.
This guide is built around the part that matters most in real lab handling. Getting a clear, stable solution without gelling, while keeping your process sterile and your dosing calculations consistent. If you’re new to reconstituting AOD 9604, treat this like bench protocol, not a casual prep task.
Table of Contents
- Assembling Your Essential Reconstitution Toolkit
- The AOD 9604 Reconstitution Protocol Step by Step
- Mastering Concentration and Dosage Calculations
- How to Prevent AOD 9604 Gelling
- Post-Reconstitution Storage and Handling for Stability
- Regulatory Compliance and Safe Research Practices
Assembling Your Essential Reconstitution Toolkit
AOD 9604 usually gets wasted before the chemistry fails. A rushed setup, cold supplies, or the wrong syringe can turn a straightforward reconstitution into a cloudy, stubborn, gelled vial.
Set the bench correctly first. That decision protects sterility, keeps your measurements tight, and reduces the handling mistakes that trigger gelling.
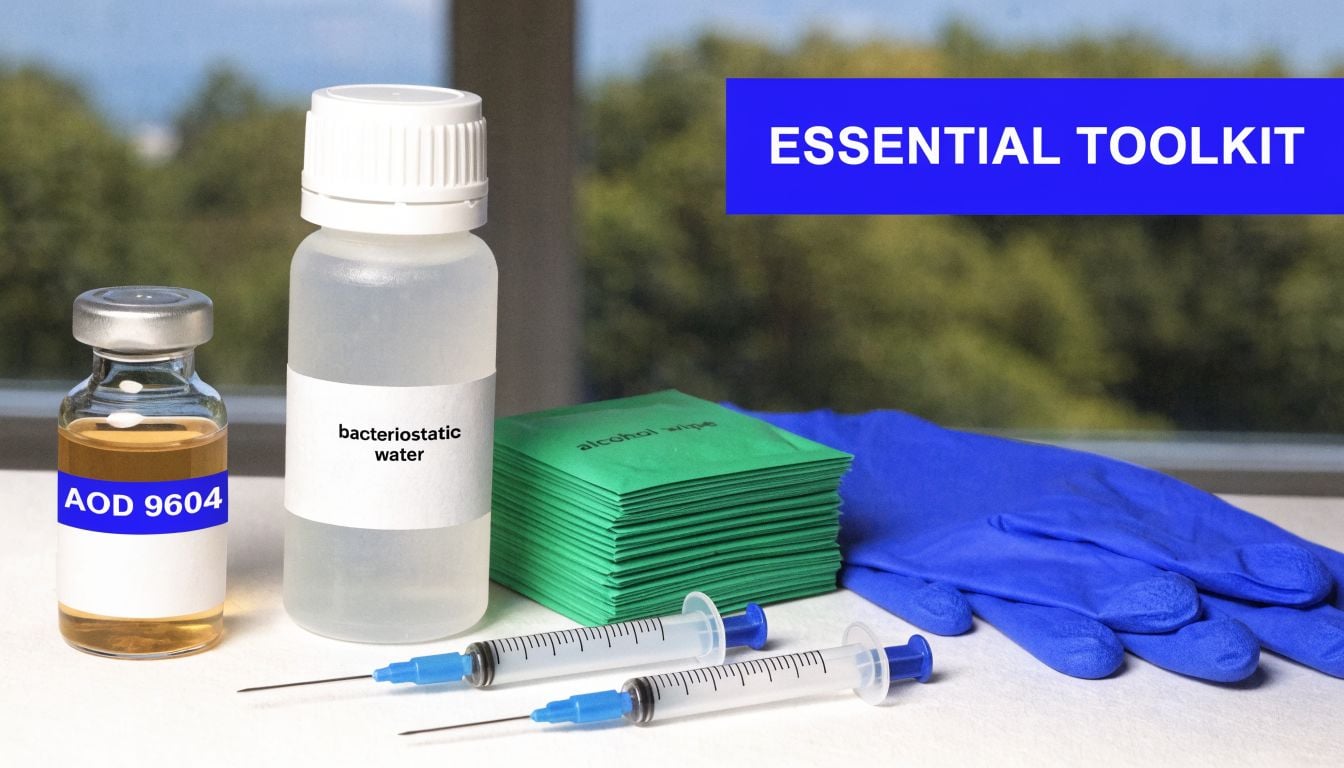
Build the workspace before you open anything
Use a clean, dry surface with enough room to keep each item separated and visible. Crowding the bench leads to crossed hands, extra stopper contact, and preventable contamination. It also encourages rushed movements, which matter with AOD 9604 because rough handling increases the chance of poor dissolution.
Lay out every item in order of use before the vial is punctured. Keep the peptide vial upright, the diluent nearby, and your syringes capped until the moment you need them.
At minimum, prepare:
- AOD 9604 lyophilized vial: Check that the stopper is seated properly and the cake or powder looks dry, intact, and free of obvious collapse.
- Bacteriostatic water: Use a diluent appropriate for repeated sterile withdrawals. Room-temperature diluent is the safer starting point for this peptide because cold liquid is a common setup for gelling problems.
- Sterile transfer syringe: Reserve one syringe for drawing and adding diluent so you can control the flow and keep the process clean.
- U-100 insulin syringes: These make later measurement easier because the graduations are readable at small volumes.
- Alcohol prep pads: Disinfect the vial stoppers before each puncture and let the alcohol dry fully.
- Nitrile gloves: Gloves reduce direct contact contamination, but only if you avoid touching nonsterile surfaces once they are on.
- Lab label or notebook: Record the peptide amount, diluent volume, concentration, date, and any handling notes immediately.
One missing tool can ruin the run. If you have to stop halfway to find a syringe or label, the vial stays exposed longer and the risk of contamination climbs.
Choose tools that support precise handling
AOD 9604 work is unforgiving at small volumes. Minor measuring errors shift concentration enough to create bad records, inconsistent draws, or unnecessary repeat work.
Choose a transfer syringe that gives you control, not speed. A syringe that is too large makes it harder to add diluent gently along the vial wall. A syringe that is too small may force multiple transfers, which means more punctures and more opportunities to introduce contamination or disturb the powder.
The insulin syringe matters for the same reason. Clear unit markings make repeatable withdrawals easier once the vial is fully reconstituted. Good visibility sounds basic, but poor readability is one of the fastest ways to drift off target during routine handling.
A practical setup usually includes:
- One syringe used only for diluent transfer
- Separate insulin syringes used only for measured withdrawals
- Fresh needles as needed to avoid dull punctures and stopper damage
- A written record created before the first draw, not after
Researchers new to peptide handling often focus on the vial and ignore the bench conditions around it. In practice, the environment usually causes the first mistake. Controlled setup, room-temperature materials, clean stoppers, and the right syringe combination give AOD 9604 the best chance to dissolve cleanly without turning into an avoidable gel.
The AOD 9604 Reconstitution Protocol Step by Step
AOD 9604 is often lost at the reconstitution stage, not because the materials were bad, but because the peptide was hydrated too fast, with cold diluent, or with too much force. Gelling usually starts there.
The goal is simple. Hydrate the lyophilized powder evenly, keep mechanical stress low, and give the peptide time to dissolve on its own.

Prepare the vial and diluent
Place the peptide vial and bacteriostatic water on a clean bench before starting. If the diluent came from refrigeration, let it reach room temperature first. Cold bacteriostatic water increases the chance of poor dissolution and can push AOD 9604 toward a stringy or gelled appearance during hydration.
Swab both stoppers with alcohol and let them dry fully. Dry time matters. Puncturing a wet stopper can carry alcohol into the vial, and touching the stopper again defeats the cleaning step.
Check the vial before you begin. The lyophilized cake should look intact and dry. If it already looks collapsed, damp, or stuck heavily to the glass, use extra care during the first milliliters of diluent because that material can clump faster.
Add the diluent slowly and along the vial wall
The first contact between water and peptide determines how the rest of the vial behaves. Fast injection creates turbulence. Turbulence traps bubbles, hits the powder directly, and raises the odds of clumping or gelling.
Use a steady hand and follow this sequence:
- Draw the planned volume of bacteriostatic water carefully to limit bubbles in the syringe.
- Insert the needle through the stopper with the tip aimed at the inner glass wall, not at the peptide cake.
- Start the flow slowly so the first portion of diluent runs down the wall and wets the powder gradually.
- Keep the stream off the powder bed for the entire transfer. Direct impact is one of the easiest ways to ruin the texture of the solution.
- Finish the transfer at the same controlled pace rather than speeding up near the end.
This is a technique step, not just a cleanliness step. AOD 9604 responds better to gentle hydration than to speed. In practice, slower addition takes only a little longer and prevents the much larger cost of a failed vial.
A quick visual reference can help if you want to compare your hand position and vial angle during setup:
If the diluent strikes the powder directly, the vial can turn cloudy, stringy, or gelatinous before the peptide has a fair chance to dissolve.
Let the peptide dissolve with minimal agitation
Once the full volume is in the vial, set the pace even lower. Shaking is a common mistake. It introduces foam, increases shear stress, and makes a borderline reconstitution worse.
Use gentle swirling or a light rolling motion between the fingers. Then wait. Some vials clear quickly. Others need several minutes of quiet rest before the last visible material dissolves.
Watch for these visual cues:
- Clear solution: the target result
- Fine bubbles only: usually harmless if they settle
- Cloudiness or strings: a sign the peptide needs more time and less handling
- Thickened or gel-like movement: a warning that temperature, injection speed, or direct powder impact may have disrupted the reconstitution
Do not keep mixing just because the vial is not instantly clear. Repeated agitation often makes the texture worse. In the lab, patience here saves more material than any rescue attempt later.
Confirm the endpoint before first use
The vial is ready only when the solution looks uniform and free of visible undissolved material. Hold it up to the light and rotate it slowly. You are checking for clarity, consistency, and any strands clinging to the glass.
If the solution still looks uneven, let it rest again and return to gentle swirling only if needed. Force rarely fixes AOD 9604. Controlled hydration does.
Mastering Concentration and Dosage Calculations
A vial can reconstitute perfectly and still become a failed batch on paper. The usual mistake is simple. The researcher remembers the vial contains 5 mg, forgets the exact final volume, and draws by habit instead of by concentration.
For AOD 9604, that kind of shortcut creates two problems at once. Dose accuracy slips, and troubleshooting becomes harder if the vial later shows instability. When you are trying to prevent waste, every vial needs a clear concentration record from the start.
Start with the only formula that matters
Convert the vial strength to micrograms, then divide by the final reconstitution volume.
For a 5 mg vial:
- 5 mg = 5,000 mcg
- Concentration = 5,000 mcg ÷ total mL added
That gives you a working concentration you can use for every draw. No guessing. No mental math at the bench.
Two practical setups for a 5 mg vial
The two setups below are common because they balance precision differently:
| Total Diluent Added | Resulting Concentration | Volume for 300 mcg Dose |
|---|---|---|
| 2.0 ml | 2,500 mcg/ml | 0.12 ml |
| 3.0 ml | 1,667 mcg/ml | 0.18 ml |
The trade-off matters.
A 2.0 ml reconstitution gives a more concentrated vial, so each dose requires a smaller draw. That reduces injection volume, but it also makes small measuring errors more significant. A 3.0 ml reconstitution gives a less concentrated solution, which is often easier to measure consistently on a U-100 insulin syringe. The downside is a larger draw volume for the same dose.
In practice, researchers who struggle with tiny volume accuracy often do better with the more diluted setup. Researchers who need the smallest possible draw may prefer the stronger one. Neither is automatically better. The correct choice is the one your measuring tools and handling technique can reproduce reliably.
Convert concentration into syringe units
A U-100 insulin syringe measures 100 units per 1 ml. That means:
- 1 unit = 0.01 ml
- 10 units = 0.1 ml
- 20 units = 0.2 ml
Now apply that to the reconstituted vial.
If the vial concentration is 2,500 mcg/ml, then:
- 0.1 ml = 250 mcg
- 0.12 ml = 300 mcg
- 10 units = 250 mcg
- 12 units = 300 mcg
If the vial concentration is 1,667 mcg/ml, then:
- 0.1 ml = about 167 mcg
- 0.18 ml = about 300 mcg
- 10 units = about 167 mcg
- 18 units = about 300 mcg
Write these conversions down once. Then label the vial and the study log the same way every time. That step prevents one of the most common bench errors: switching to a fresh vial and forgetting that it was reconstituted to a different final volume than the last one.
Use a repeatable calculation method
A simple three-step method keeps the math clean:
-
Convert mg to mcg
5 mg = 5,000 mcg -
Divide by total volume
5,000 mcg ÷ 2.0 ml = 2,500 mcg/ml -
Divide target dose by concentration
300 mcg ÷ 2,500 mcg/ml = 0.12 ml
That final number is the draw volume.
This matters more than it looks. Researchers often focus on the chemistry of reconstitution and treat the math like an administrative task. It is not. A concentration error can make a properly prepared vial functionally useless for the protocol.
Calculation habit: Label the vial immediately after reconstitution with the total volume, calculated concentration, and date. Do not trust memory once multiple vials are in cold storage.
One more practical point. If you change your reconstitution volume from vial to vial, document the reason. Consistency helps dosing accuracy, and it also helps isolate what caused a problem if one batch later behaves differently from another.
How to Prevent AOD 9604 Gelling
You add the diluent, the cake seems to wet, and within minutes the vial turns stringy or thick. At that point, recovery is unreliable, and the batch often has to be discarded. Gelling is the most common way AOD 9604 gets wasted during reconstitution, and it usually starts in the first few seconds of liquid contact.

Why AOD 9604 gels so easily
AOD 9604 is sensitive at the hydration stage. The main problem is not the final volume by itself. The problem is how the first portion of diluent hits the peptide and how quickly the local environment changes inside the vial.
In practice, gelling usually traces back to three avoidable errors:
- Diluent that is too cold: Cold liquid can create an abrupt hydration shock.
- A hard, fast stream into the powder: That creates high-concentration pockets before the material has time to dissolve evenly.
- Aggressive mixing: Shaking, flicking, or repeated forceful inversion can push a fragile peptide toward clumping instead of clear dissolution.
This is why two researchers can use the same vial, the same total volume, and get different results. Technique changes the outcome.
Handling habits that reduce the risk
The best prevention is controlled hydration. Every step should reduce local stress inside the vial.
Start with room-temperature diluent. If bacteriostatic water has been refrigerated, let it sit until it is no longer cold to the touch. Then inject slowly with the needle bevel aimed at the inner glass wall, not directly into the peptide cake. The goal is to let the liquid run down the wall and spread gradually.
After the first portion is added, pause. Let the powder wet and settle before adding more. Once the full volume is in, use a gentle swirl only if needed. Do not shake the vial. Do not try to force clarity in seconds.
A practical bench rule helps here: if the vial still shows undissolved material, give it time before touching it again. Rushed handling causes more failures than slow dissolution.
What to watch for during reconstitution
A properly reconstituting vial usually moves from dry cake to partially wetted material, then to a clear solution. A problem vial often shows warning signs early. You may see stringiness, cloudy threads, thick patches near the bottom, or material that seems to clump where the first stream landed.
Stop and assess if that happens. More agitation rarely fixes the underlying issue. It usually makes the texture worse.
For that reason, the safest workflow is consistent and boring:
- Bring the diluent to room temperature.
- Sanitize the stopper and use sterile technique.
- Inject down the vial wall in a slow, controlled stream.
- Let the vial rest between additions if needed.
- Swirl lightly, then wait for full dissolution before any further handling.
When a mixed-diluent approach makes sense
Some labs can reconstitute AOD 9604 with bacteriostatic water alone if technique is tight and temperature is controlled. Other labs run into repeat gelling with certain lots or under certain workflows. In those cases, a mixed-diluent protocol may be worth evaluating.
Acetic acid is sometimes used as part of that adjustment because changing the reconstitution environment can improve peptide solubility behavior. The trade-off is extra complexity. You now have another reagent, another measurement step, and less room for casual technique. If a lab adopts that approach, it should be standardized, documented, and repeated the same way every time.
If your current method gives a clear solution without repeated failures, keep it simple. If gelling keeps happening despite careful room-temperature handling, slow wall injection, and gentle mixing, then a revised solvent system is a reasonable troubleshooting step, as noted earlier.
Post-Reconstitution Storage and Handling for Stability
The vial looked perfect when it was mixed. Two days later, the solution is cloudy, the stopper has been pierced half a dozen times, and nobody is sure how long it sat on the bench between draws. That is how good material gets lost after a technically correct reconstitution.
Post-reconstitution stability is mostly a handling problem. Once AOD 9604 is in solution, the goal is to limit the stresses that push a clean, usable preparation toward degradation, contamination, or a visible change that makes the sample questionable. Labs that struggle with gelling during reconstitution often make a second mistake here by exposing the vial to repeated temperature swings. A peptide that was difficult to dissolve should be handled even more carefully after mixing.
Keep storage conditions boring and consistent
Store the reconstituted vial under refrigeration at a stable, cold temperature. Protect it from light. Keep it upright when possible, and do not freeze the solution unless your protocol specifically validates that approach.
Consistency matters more than people expect. A vial that warms up on the bench several times a day and then goes back into the refrigerator is under repeated stress. That does not guarantee failure, but it does increase the chance of appearance changes, concentration drift from handling loss, and avoidable questions about sample quality.
A practical storage routine looks like this:
- Refrigerate the vial promptly after reconstitution
- Minimize time at room temperature during each use
- Store away from direct light
- Keep the cap, stopper, and outer container clean and dry
- Avoid repeated freeze-thaw cycles
If a study requires frequent access, aliquoting can be the cleaner option. That reduces how often the primary vial is pierced and helps limit the warm-cold cycling that can contribute to instability.
Treat every draw as a contamination event waiting to happen
The stopper is the weak point after reconstitution. Every needle entry creates another chance to introduce contamination or compromise the closure.
Use the same routine every time. Swab the stopper with alcohol and let it dry fully before puncturing. Use a new sterile syringe and needle for each draw. Return the vial to cold storage without leaving it out while other tasks are finished.
Before withdrawing any solution, inspect the vial under good light. Look for cloudiness, stringiness, surface film, particles, or a change in how the liquid moves along the glass. Those are not details to dismiss, especially in a guide focused on preventing wasted peptide from solubility and handling failures. If the solution no longer matches its original appearance, quarantine it until the issue is reviewed.
Good records help here. Label the vial with the reconstitution date, diluent used, and final concentration.
Regulatory Compliance and Safe Research Practices
AOD 9604 should be handled with the same seriousness you’d apply to any sensitive research compound. That means precise documentation, controlled storage, and clear separation between research use and anything outside that scope.
Quality handling supports quality data
A sloppy reconstitution doesn’t just waste material. It undermines consistency across the entire protocol.
Safe practice usually comes down to a short list:
- Document every vial clearly: Record lot details, reconstitution date, and final concentration.
- Keep technique consistent: Don’t change diluent volume or handling style halfway through a study unless the protocol itself changes.
- Treat sterility as part of data quality: Contaminated handling compromises more than appearance.
- Use only research-appropriate supplies and compounds: High-purity materials and traceable documentation support cleaner work.
That standard protects the sample, the procedure, and the credibility of the result.
For researchers who want USA-made, high-purity compounds supported by COAs and batch documentation, Peptide Warehouse USA is worth exploring. Learn more about their research peptide catalog and sourcing standards if you’re building a more dependable lab supply workflow.






Leave a comment