Peptide Purity Testing: A Guide for Researchers
You’re probably looking at a peptide product page or a Certificate of Analysis right now, trying to answer a simple question that turns out not to be simple at all. Is this batch clean enough for your work, or does the label only look reassuring?
That’s where peptide purity testing stops being a paperwork exercise and becomes part of experimental design. If you run cell assays, receptor work, stability studies, or formulation experiments, the difference between a trustworthy batch and a questionable one can show up as noisy data, shifting dose response curves, unexplained variability, or a project that refuses to reproduce.
Most researchers get told to “check the COA.” Few are taught how to read one critically. A key skill is knowing what the purity number means, what it doesn’t mean, and which raw data points reveal problems before they ruin a study.
Table of Contents
- Why Peptide Purity Is Critical for Your Research
- Understanding Common Peptide Purity Testing Methods
- A Step-by-Step Analytical Workflow
- How to Read a Certificate of Analysis COA
- Defining Quality and Acceptance Criteria
- Recommended Testing Strategies for Research Labs
- Frequently Asked Questions About Peptide Purity
Why Peptide Purity Is Critical for Your Research
Peptide purity is the foundation of reliable data. If the material in your vial contains related fragments, deletion sequences, truncations, residual salts, or biologically active contaminants, your assay may be measuring more than the target peptide.

Small impurities create big experimental problems
The common mistake is treating impurities as passive background. They often aren’t. In peptide work, an impurity can bind weakly to the same receptor, interfere with transport, alter solubility, or change how a sample behaves during storage and reconstitution.
For assay-driven research, that matters immediately. GenScript notes that peptide purity grades directly impact assay performance, with more than 95% purity required for enzyme kinetics or cell-based bioactivity studies, and that crude peptides below 70% purity can introduce deletion variants that bind non-specifically and skew EC50 values by 10-fold to 50-fold in assays involving peptides such as GHK-Cu or BPC-157 (GenScript peptide purity guidance).
Practical rule: If your experiment depends on a quantitative biological response, purity isn’t a luxury specification. It’s part of the assay method.
A peptide batch can also trigger confusion in vivo or in complex cell systems when low-level contaminants provoke unintended responses. That doesn’t always look dramatic. Sometimes it appears as inconsistent replicate behavior, unexpected baseline movement, or a result that can’t be repeated with the next lot.
Purity affects reproducibility, not just appearance
A clean chromatogram gives you more than a nice number for a purchasing file. It gives you confidence that the test article is stable enough, specific enough, and consistent enough for the question you’re asking.
That’s why experienced labs think about peptide quality in terms of risk:
- For screening work: lower purity may be acceptable if the goal is early directional information.
- For mechanistic studies: related impurities can distort receptor, signaling, or enzyme data.
- For comparative studies: lot-to-lot consistency matters as much as the headline purity claim.
- For long projects: even small batch differences can accumulate into conflicting datasets.
Researchers in weight loss, repair, anti-aging, and muscle growth models often focus on peptide name first. The better habit is to focus on analytical trustworthiness first. If the batch isn’t well characterized, the biology becomes harder to interpret.
Understanding Common Peptide Purity Testing Methods
A good peptide test panel works like a team, not a single instrument. Each method answers a different question. One separates components, another checks molecular identity, and another helps confirm how much true peptide is present.
HPLC separates the mixture
Think of high-performance liquid chromatography, or HPLC, as the sorting step. The sample moves through a column, and different compounds travel through that system differently based on their chemical properties. The detector records those separated components as peaks.
In reversed-phase HPLC, purity is calculated from the main peak area divided by the total integrated peak area detected by UV absorbance. Vanguard Laboratory explains the calculation directly: Purity (%) = (Area_main / Total_area) × 100. Their example shows a main peak of 2,500,000 AU and total peaks of 2,575,000 AU, which gives 97.1% purity, with the remaining 2.9% representing impurities such as deletion sequences or truncations (Vanguard Laboratory guide to HPLC testing for peptides).
That formula is simple. Interpreting it correctly is harder.
A few points matter when you read an HPLC result:
- The main peak should dominate clearly. A strong central peak with only minor secondary peaks is easier to trust.
- Method details matter. Wavelength, gradient, column type, and temperature affect separation quality.
- Co-elution is real. Two compounds can overlap and look like one cleaner peak unless another method catches the problem.
A purity number without the underlying chromatogram tells you less than most buyers assume.
LC-MS confirms identity
If HPLC tells you how many detectable components are in the mixture, LC-MS helps tell you whether the main one is the peptide you ordered. The chromatography separates. The mass spectrometer measures mass-to-charge patterns and helps confirm molecular identity.
A clean-looking chromatogram isn’t enough on its own. A single dominant peak can still represent the wrong compound, a modified version, or a co-eluting impurity. Mass confirmation reduces that risk.
In practice, this is why many peptide COAs pair HPLC with MS. The two methods answer different questions:
| Method | Main job | What it helps you detect |
|---|---|---|
| HPLC | Separation and relative purity | Visible impurity profile |
| LC-MS | Identity confirmation | Expected molecular mass and related species |
| CE or other orthogonal methods | Alternative separation behavior | Polar or difficult impurities HPLC may miss |
AAA and other orthogonal methods close the gaps
Researchers often stop at “identity plus purity,” but that leaves an important blind spot. Purity is not the same as content. A vial can contain a highly pure peptide fraction while still carrying water or counterions that affect the true amount of target material.
Amino acid analysis, often shortened to AAA, helps address that. It hydrolyzes the peptide and checks residue composition, which is useful when you need confidence in actual composition rather than just chromatographic appearance.
Other orthogonal tools also matter:
- Capillary electrophoresis: useful when a peptide is highly hydrophilic or behaves poorly in standard reversed-phase systems.
- UV spectroscopy: supports concentration and detection workflows.
- MALDI-TOF mass spectrometry: helps with mass confirmation in many labs.
- NMR: used more selectively when structural confirmation is needed.
The core lesson is straightforward. No single method tells the whole story. Good peptide purity testing combines separation, identity confirmation, and, when the work demands it, content verification.
A Step-by-Step Analytical Workflow
Most new researchers see only the last page of the report. In the lab, the result comes from a sequence of controlled steps. If one of those steps is weak, the final purity claim becomes less meaningful.
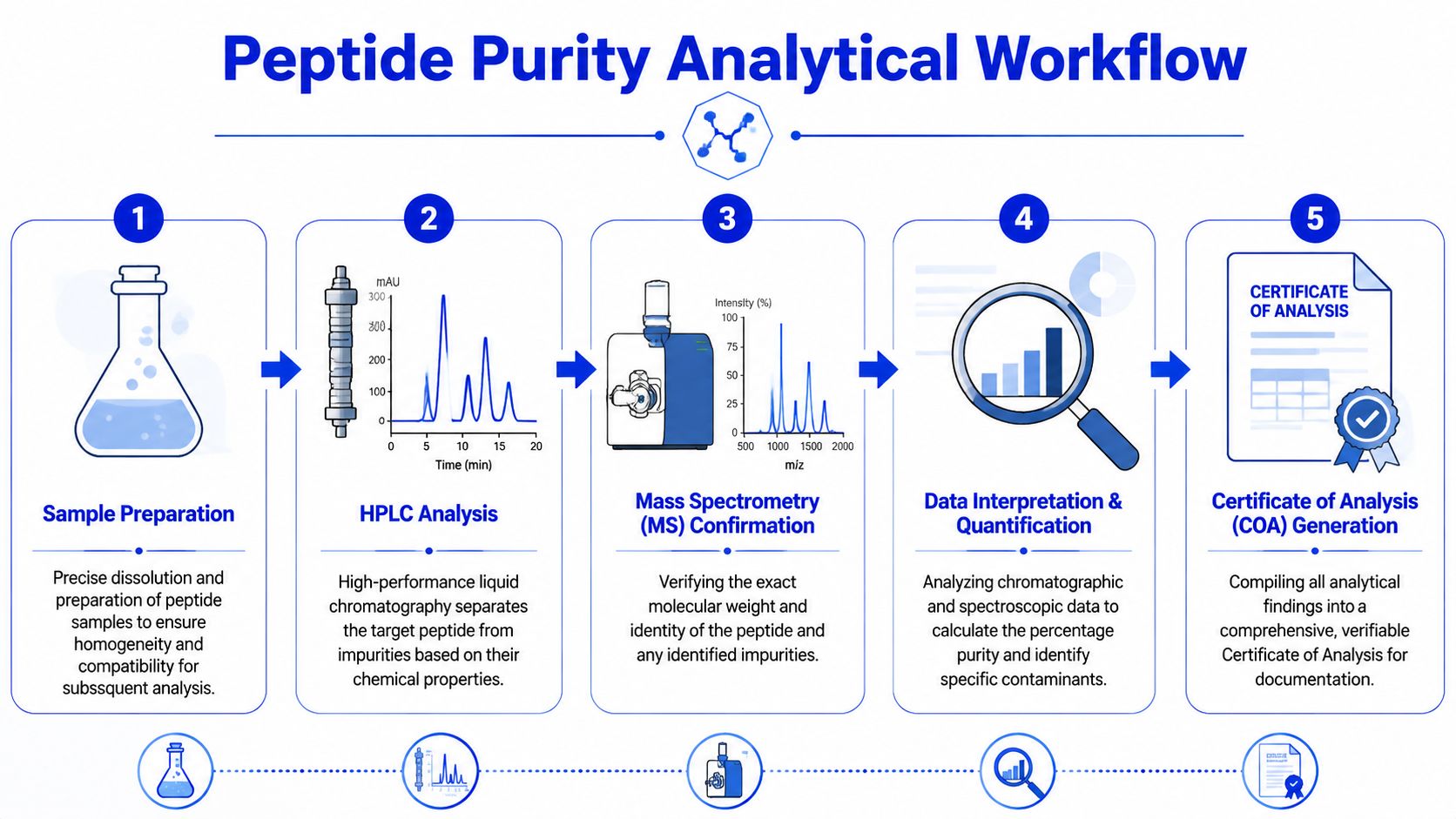
What happens from vial to result
A typical peptide purity workflow looks like this:
-
Sample preparation
The lyophilized material is dissolved carefully in a solvent system that matches the analytical method. Good dissolution matters because partially dissolved material can create misleading results. -
Chromatographic separation
The prepared sample goes into the HPLC or UHPLC system. The method separates the main peptide from related impurities, synthesis byproducts, and degradation products. -
Identity confirmation
The main peak, and sometimes impurity peaks, are checked by mass spectrometry to verify molecular identity. -
Data integration
The analyst integrates the chromatogram, assigns peaks, reviews signal quality, and calculates the purity from relative peak area. -
Reporting
The final output becomes a COA with batch details, method information, chromatograms, and identity data.
Why sequence matters in the workflow
This order isn’t arbitrary. You can’t interpret the mass result properly if the sample was poorly prepared. You can’t trust the purity calculation if the chromatography didn’t resolve the mixture. And you can’t compare lots meaningfully if reporting formats change from batch to batch.
A disciplined workflow also improves traceability. When a later batch behaves differently, the lab can ask the right questions:
- Was the sample dissolved the same way?
- Did the chromatographic method stay consistent?
- Did the same impurity pattern reappear?
- Did mass confirmation support the same identity call?
The most trustworthy COA is the end product of a stable analytical workflow, not just a polished PDF.
That’s why experienced buyers ask for more than a summary percentage. They want evidence that the route from sample receipt to final report was technically sound.
How to Read a Certificate of Analysis COA
A COA should help you verify quality. Too often, it only gives you a headline. The difference is whether the document includes enough raw analytical context to support the conclusion.

Start with the batch details
Before you even look at the purity line, check whether the document is specific to the batch in hand. A serious COA should connect clearly to the lot number, the peptide name, and the date of testing.
If you archive a lot of supplier paperwork, tools that help pull structured fields from PDFs can save time. For labs building internal QC records, PDF AI’s complete extraction guide is a useful reference for turning analytical PDFs into searchable data.
A few opening checks catch a surprising number of problems:
- Batch traceability: the lot on the vial should match the lot on the COA.
- Method visibility: the document should identify at least the analytical methods used.
- Test specificity: a generic “representative” COA is weaker than a batch-specific report.
Read the chromatogram before the summary line
Most buyers jump straight to the stated purity. Don’t. Read the chromatogram first.
A more trustworthy chromatogram usually has a dominant main peak with smaller, separated impurity peaks. A less convincing one may show broad shoulders, unresolved clusters, strange baseline movement, or a tiny image that makes interpretation impossible.
Independent testing matters because vendor claims and actual analytical results don’t always match. Resolve Mass highlights this problem with Finnrick’s database of 7,164 samples from 205 vendors, showing frequent gaps between claimed purity, often above 98%, and measured results in independent testing (Resolve Mass analysis of peptide purity testing in the United States).
That finding changes how you should read a COA. Treat it as evidence to inspect, not a claim to accept.
Signs a COA is easier to trust
| What you see | Why it matters |
|---|---|
| Clear lot number match | Confirms the document belongs to the actual batch |
| Raw chromatogram included | Lets you inspect peak shape and impurity pattern |
| MS identity data present | Supports that the main peak is the expected peptide |
| Third-party attribution | Reduces conflict of interest |
| Additional contamination screens | Improves confidence for sensitive work |
Red flags worth pausing on
- Only a purity percentage appears, with no chromatogram.
- The image is too small to read, which prevents independent review.
- No lab identity is shown, so you don’t know who performed testing.
- No impurity context is provided, only a passing claim.
- The COA is reused across lots, which defeats batch-specific verification.
To see another walkthrough of what researchers often look for when reviewing peptide documents, this short video is a helpful visual reference:
Check identity, content, and independent verification
A complete reading of the COA means separating three ideas that often get blurred together.
First is identity. Does the mass data support the expected peptide?
Second is purity. Does the chromatogram show a dominant main component with limited detectable related impurities?
Third is content. How much true target peptide is present once water and counterions are considered?
Many researchers stop at the second question. That’s exactly where poor purchasing decisions start. If the supplier never addresses content, you may calculate doses from a number that looks better than the material really is.
Don’t ask only “What purity did they report?” Ask “What raw data supports it, and what did they leave out?”
Defining Quality and Acceptance Criteria
Not every project needs the same grade of material. The right standard depends on what the peptide is being used for and how sensitive the readout is.
Match the grade to the experiment
Industry benchmarks distinguish between research and therapeutic expectations. Research peptides typically require more than 90% purity, while therapeutic-grade peptides for preclinical and clinical applications demand at least 98% purity (industry benchmarks summarized by Resolve Mass).
That doesn’t mean every experiment should chase the highest available number. It means your acceptance criteria should match the scientific risk of impurity interference.
For example:
- Exploratory screening can tolerate more uncertainty than quantitative potency work.
- Cell-based bioactivity studies need tighter control because impurities can alter biological readouts.
- Preclinical support work needs stronger documentation, especially when the data may guide later decisions.
If your work is sensitive to inflammatory artifacts, contamination controls matter too. A plain-language explainer on bacterial endotoxin is useful background for teams that review peptide COAs but don’t routinely interpret microbiological risk.
Peptide purity grades and common applications
Here’s a practical way to think about acceptance criteria.
| Purity Level | Common Impurities | Recommended Application |
|---|---|---|
| Crude or low purity | Deletion sequences, truncations, synthesis byproducts | Early screening or non-quantitative exploratory work |
| Above 90% | Reduced but still relevant related impurities | General research use where moderate impurity risk is acceptable |
| Above 95% | Lower impurity burden, stronger consistency | Enzyme kinetics and cell-based bioactivity studies |
| At least 98% | Minimal detectable related impurities with stronger documentation expectations | Preclinical or clinical-facing analytical contexts and high-confidence quantitative research |
This table is a decision aid, not a shortcut. The cleaner the peptide needs to be, the more important it becomes to review supporting raw data, not just the final line item.
Recommended Testing Strategies for Research Labs
Most labs don’t need to independently retest every incoming vial. They do need a consistent system for deciding when verification is worth the cost and effort.
Use a trust but verify model
Start by treating supplier documentation as useful, but not self-authenticating. A reliable workflow in the lab usually includes an intake review for every batch and a deeper review for batches tied to critical experiments.
Your routine can be simple:
- Screen each COA for completeness: batch match, chromatogram, identity data, and contamination screens where relevant.
- Hold a reference copy internally: save PDFs in a searchable folder by lot number and project.
- Compare lots over time: watch for changes in peak shape, impurity pattern, or reporting style.
- Escalate when biology changes: if a new batch behaves differently, review the analytics before you blame the assay.
When to escalate to outside testing
One of the most overlooked issues is the gap between purity and actual usable amount. GenScript’s quality guidance states that true target peptide = gross weight × net peptide × HPLC purity, and notes that suppliers who report purity without net peptide content can inflate perceived yield by 10% to 30% (GenScript AccuPep quality guidance).
That formula has practical consequences. If your dose calculations assume the label weight equals target peptide weight, you may be dosing incorrectly even when the peptide looks “high purity.”
Outside testing is worth considering when:
- A project is expensive or long-running
- A new supplier is being qualified
- A batch gives inconsistent biological behavior
- The COA lacks raw data or content transparency
- You need independent records for internal quality systems
The best labs don’t verify everything. They verify the batches that carry the most scientific risk.
Frequently Asked Questions About Peptide Purity
Is purity the same as peptide content
A new researcher often sees “98% purity” on a COA and assumes 98% of the vial is usable target peptide. That reading is incomplete.
Purity usually refers to the proportion of the detected signal assigned to the target peak compared with other detected peaks under a specific analytical method. Peptide content answers a different lab question: how much actual target peptide mass is present after you account for water, salts, and counterions. In practice, purity helps you judge chemical cleanliness, while content helps you calculate dose.
A batch can look clean by HPLC and still deliver less target peptide per milligram than your experiment assumes.
Why do counterions matter
Counterions such as TFA or acetate add weight. Water does too. Neither is the peptide sequence you designed the experiment around.
That matters during reconstitution. If you calculate concentration from gross vial weight alone, your molar dose can be off, sometimes enough to confuse assay interpretation. The effect is similar to weighing a sample in a wet container. The balance gives a real number, but not the number you need.
When you read a COA, check whether the supplier distinguishes gross weight, net peptide content, and purity. If those terms are blurred together, treat the stated yield cautiously.
Can a high-purity peptide still cause poor results
Yes. High purity reduces one source of uncertainty, but it does not clear the batch of every other risk.
A peptide may have acceptable purity and still present problems from oxidation, deletion sequences below the reporting threshold, residual solvents, storage damage, or weak batch documentation. Method choice matters too. A single HPLC method can miss impurities that separate poorly under those conditions.
This is why experienced analysts do not stop at the final percentage. They inspect the chromatogram, identity data, and lot-specific notes to see whether the reported result matches the raw evidence.
What purity level should I choose
Match the purity requirement to the decision your assay must support.
For early exploratory work, a lower specification may be acceptable if the biology is tolerant of some uncertainty. For receptor studies, quantitative cell work, binding experiments, or any project where small shifts in response matter, tighter purity and better supporting data usually save time.
A simple screening rule works well:
- How sensitive is the assay to small chemical differences?
- How expensive is a wrong conclusion?
If one questionable lot can cost you a repeat study, choose stricter acceptance criteria and stronger documentation.
Why should I ask for raw chromatograms
The purity percentage on a COA is a summary. The chromatogram is the underlying record.
Reading it does not require expert-level chromatography training. Start with a few practical checks. Is there one dominant peak at the expected retention time? Are there smaller peaks nearby that were excluded or left unidentified? Is the baseline stable, or noisy enough to make integration uncertain? Do the peak shapes look sharp and consistent, or broad and irregular?
Those details help you spot red flags that a headline purity number can hide. A clean-looking percentage with a messy trace deserves follow-up questions.
Is third-party testing always necessary
No. It is a risk-based decision.
Independent testing makes the most sense when the project is expensive, the supplier is new, the lot will support publication-quality conclusions, or the COA leaves unanswered questions. It is also useful when two lots behave differently and you need to determine whether the problem started in the chemistry or in the biology.
If your internal review of the raw data raises doubt, outside testing gives you a second set of eyes and a separate analytical record.
If you want research peptides backed by transparent batch documentation, including third-party COAs and supporting quality records, explore Peptide Warehouse USA. You can review available options, compare formats, and learn more about a US-made supply chain built for laboratory and analytical use.






Leave a comment