Solid Phase Peptide Synthesis: A Researcher’s Guide
You’re likely here because you need a peptide you can trust. Not just a sequence on a label, but a material with the purity, identity, and batch consistency that won’t distort your research.
That’s where solid phase peptide synthesis matters. If you work with peptides for healing and repair research, anti-aging studies, metabolic research, or analytical method development, the way a peptide is made directly affects the confidence you can place in every result that follows. A clean chromatogram, a credible COA, and reproducible handling all begin at the synthesis bench.
Most articles stop at a simple definition. That misses the central point. The chemistry choices made during synthesis, from resin selection to deprotection strategy to purification, shape the final peptide a researcher receives.
Table of Contents
- The Journey of a Peptide From Concept to Creation
- The Core Principles of Solid Phase Peptide Synthesis
- Choosing Your Strategy Fmoc versus Boc Chemistry
- The Essential Building Blocks Resins Linkers and Reagents
- The SPPS Workflow From Manual Synthesis to Automation
- Ensuring Purity and Identity Purification and Analysis
- Troubleshooting and Sourcing High-Quality Peptides
The Journey of a Peptide From Concept to Creation
A peptide starts as an idea on paper. A researcher chooses a sequence, maybe a short signaling peptide, maybe a modified analog, and then faces a practical question. How do you assemble that chain with enough control that the final material is worth testing?
That question used to be much harder to answer. Before modern methods, peptide synthesis was slow, purification-heavy, and frustrating for longer chains. Every step could generate byproducts that had to be separated before the next amino acid could be added.
R. Bruce Merrifield changed that. He invented solid-phase peptide synthesis in 1959, published the method in 1963, and built the field around one deceptively simple idea: anchor the first amino acid to an insoluble resin, then build the peptide while it stays attached. That innovation reduced synthesis time from years to days and pushed reaction efficiency to over 99.5% per cycle, according to The Rockefeller University’s profile of Merrifield and his Nobel-recognized work.
He received the 1984 Nobel Prize in Chemistry for it.
That history matters because modern peptide availability depends on it. The peptides researchers order today exist in practical form because Merrifield turned peptide assembly from a slow artisanal process into a repeatable chemical workflow.
Bench perspective: If a peptide can be made quickly but not cleanly, it still isn’t useful. Merrifield’s real achievement was making speed and cleaner handling possible at the same time.
For the researcher, the lesson is simple. A peptide’s reliability doesn’t start with packaging or shipping. It starts with whether the synthesis method controlled each amino acid addition, protected vulnerable groups at the right time, and limited the buildup of side products.
That’s why solid phase peptide synthesis sits underneath nearly every serious conversation about research peptide quality. When you understand how it works, you can read a specification sheet with very different eyes.
The Core Principles of Solid Phase Peptide Synthesis
The easiest way to understand solid phase peptide synthesis is to picture a molecule being built on a bench fixture. The resin is the fixture. The peptide chain grows from it one amino acid at a time.
That fixed position is the whole trick. Because the growing chain stays attached to a solid support, the chemist can flush away excess reagents and byproducts after each cycle without losing the peptide itself.
Why the solid support changes everything
In older liquid-phase synthesis, the peptide and the unwanted materials all stayed in solution together. That meant repeated purification steps during assembly. For short molecules, that might be manageable. For longer peptides, it becomes a bottleneck.
With SPPS, the chain remains anchored while the reaction vessel is washed. That makes the workflow faster, simpler, and far more practical for peptides longer than a few residues. CPC’s overview of why solid-phase synthesis dominates modern peptide production notes that SPPS is significantly more efficient than liquid-phase synthesis, especially for peptides over five amino acids, and that the deprotection, coupling, and washing cycle is the key reason it became the dominant method in research and pharmaceutical production.
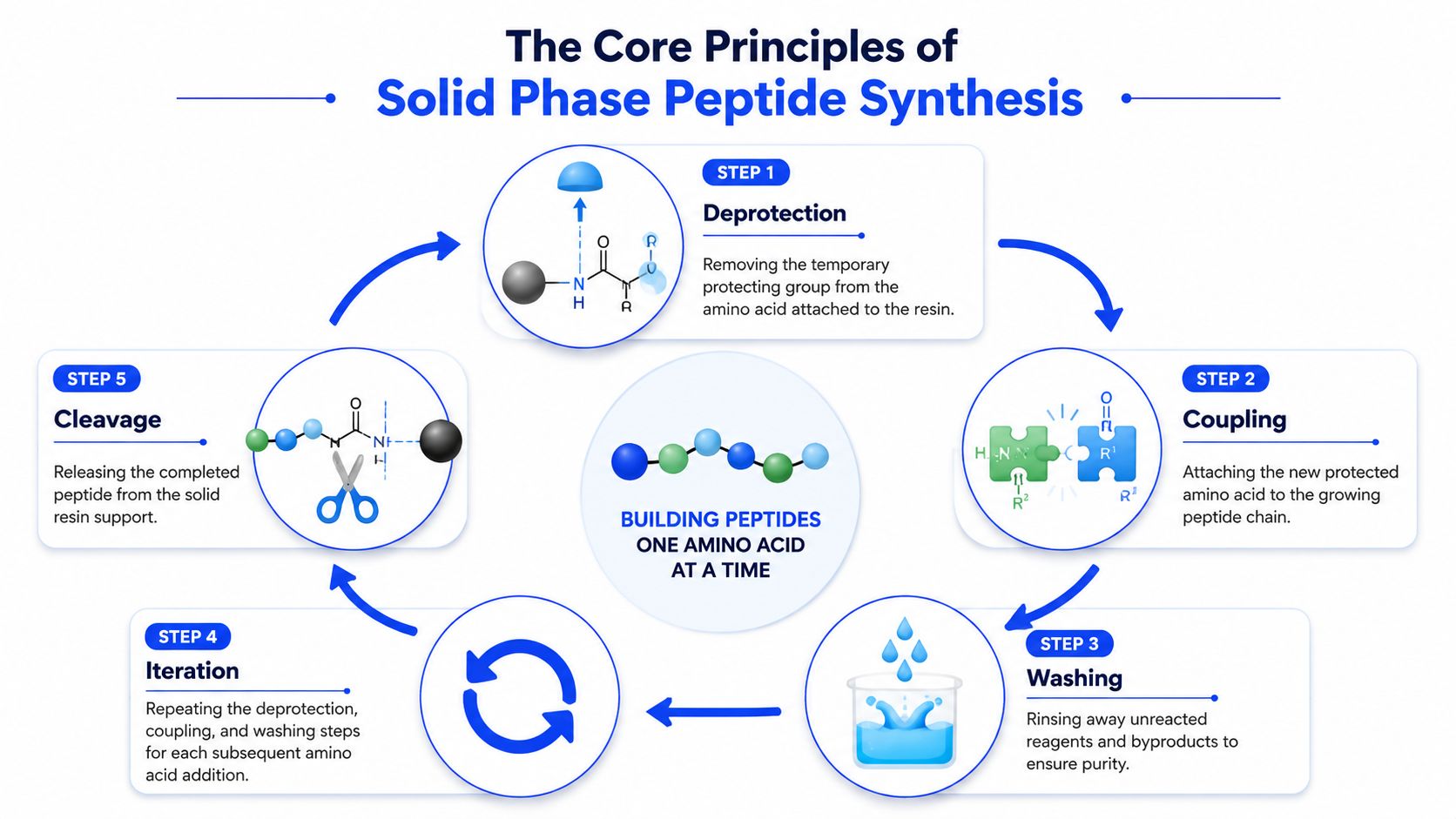
The repeating cycle that builds the chain
At the bench, the synthesis cycle looks straightforward. In practice, each step has to be executed cleanly or the final crude peptide gets messy fast.
- Anchor the first amino acid to the resin through a linker.
- Remove the temporary protecting group from the exposed amine.
- Couple the next protected amino acid onto the growing chain.
- Wash thoroughly to remove leftover reagents and side products.
- Repeat until the full sequence is assembled.
- Cleave the finished peptide from the resin at the end.
A useful analogy is building on a baseplate. The resin holds the growing structure in place so you can add the next piece, rinse away debris, and keep going without the whole assembly drifting away.
Readers often get confused about the word “protection.” It doesn’t mean the amino acid is chemically inactive everywhere. It means specific reactive groups are temporarily blocked so the peptide grows in the intended direction, one bond at a time, instead of reacting in multiple unwanted ways.
The wash step looks boring on paper. In real synthesis, it’s one of the reasons the final peptide is either manageable or full of avoidable impurities.
Another point of confusion is why SPPS remains useful even when the chemistry is repetitive. The answer is that repetition is an advantage. Standardized cycles make the process easier to optimize, monitor, and automate, which is exactly what researchers want when they need consistent material across batches.
The result is a synthesis method that’s not just clever chemistry. It’s a manufacturing logic. Each cycle is designed to protect the integrity of the next one, and that cumulative control is what eventually shows up as purity, identity, and reproducibility in the sample vial.
Choosing Your Strategy Fmoc versus Boc Chemistry
Most peptide chemists eventually frame Fmoc and Boc as two different ways of solving the same problem. Both strategies let you build a peptide stepwise on resin. They differ in how they protect and unmask the growing chain during synthesis, and those choices affect side reactions, cleavage conditions, and practical workflow.
Fmoc as the modern workhorse
In Fmoc/tBu chemistry, the temporary N-terminal protecting group is removed with base. This is one reason many labs prefer it for routine synthesis. The conditions are generally easier to handle operationally, and the strategy fits well with modern automated workflows.
The catch is that Fmoc chemistry demands discipline. The Nowick laboratory’s Fmoc SPPS practices guide states that each coupling cycle should achieve >99% yield, and it shows why small losses matter. On a 20-mer peptide, 99% yield per step gives about 82% crude purity, while 98% yield per step drops that crude purity to 60%.
That difference catches many new researchers off guard. One bad deprotection or one weak coupling doesn’t just create a minor impurity. It seeds a family of truncated sequences that follow the chain all the way to the end.
Boc as a different operating philosophy
Boc/Bzl chemistry removes the temporary protecting group with acid during each cycle, then uses much stronger conditions for final global deprotection and cleavage. It’s an older but still important strategy, especially when a sequence behaves better under that chemistry.
Boc can be a good fit for certain difficult or hydrophobic peptides. It also comes with operational demands that many labs would rather avoid, especially at small scale, because the final cleavage conditions are more specialized and less forgiving from a handling standpoint.
Here’s the practical takeaway. Fmoc is often favored for convenience and compatibility with modern instruments. Boc remains relevant when sequence behavior, side-chain issues, or process familiarity make it the better choice.
Practical rule: Don’t ask which strategy is “better” in the abstract. Ask which strategy gives the cleanest path to the peptide you actually need.
Comparison of Fmoc and Boc Synthesis Strategies
| Attribute | Fmoc/tBu Strategy | Boc/Bzl Strategy |
|---|---|---|
| Temporary N-terminal deprotection | Base-mediated removal of Fmoc | Acid-mediated removal of Boc |
| Workflow fit | Common in modern manual and automated SPPS | More specialized, often chosen for particular sequence behavior |
| Sensitivity point | Incomplete deprotection can create deleted sequences that accumulate across the synthesis | Final cleavage and global deprotection conditions are operationally demanding |
| Final purity impact | Strongly depends on maintaining very high efficiency each cycle | Strongly depends on successful final deprotection and clean handling |
| Typical lab appeal | Convenient, widely adopted, instrument-friendly | Useful when its chemistry better suits the target peptide |
| Research interpretation | Often the default strategy for many custom peptides | Better viewed as a deliberate choice, not an outdated one |
A buyer won’t always be told which chemistry produced a peptide, and that’s fine if the supplier demonstrates the outcome clearly. But as a scientist, it helps to know why one sequence may synthesize cleanly in one system and poorly in another.
When a peptide is difficult, the chemistry choice isn’t just an academic preference. It affects how much cleanup is needed later, how many truncated species appear in the crude product, and how hard the purification team has to work to deliver a reliable final lot.
The Essential Building Blocks Resins Linkers and Reagents
If amino acids are the visible part of peptide construction, resins, linkers, and reagents are the hidden engineering. These choices rarely appear in product marketing, but they heavily influence whether a peptide comes off the synthesizer cleanly or arrives at purification as a complicated mixture.

Resin as the physical platform
The resin is the solid support bead where the peptide grows. It has to do two jobs well. It must hold the peptide securely through repeated reaction cycles, and it must swell appropriately in solvent so reagents can reach the reactive sites inside the bead.
That swelling point is more important than it sounds. If solvent access is poor, some parts of the resin bead become chemically under-served. The peptide then grows unevenly across the batch, which is exactly how hard-to-remove impurity families begin.
Different resins suit different sequences. Some are chosen for general utility, others for more difficult peptides. But the principle stays the same. A resin is not passive. It shapes mixing, accessibility, and consistency.
Linkers and activators decide how cleanly the peptide is made
A linker is the molecular connection between the resin and the first amino acid. It determines how the peptide will be released at the end and what kind of peptide terminus you get after cleavage.
That means linker choice is strategic. If the linker is mismatched to the target, cleavage may be harsher than necessary or the final product may not have the desired terminal functionality.
Then there are the coupling reagents, the chemical activators that help the incoming amino acid form a peptide bond with the exposed amine on the growing chain. These are the drivers of bond formation, but they also influence side reactions if conditions aren’t tuned correctly.
A chemist usually thinks about these components together:
- Resin choice affects solvent compatibility, mixing behavior, and sequence accessibility.
- Linker design affects cleavage behavior and the final peptide form.
- Reagent system affects coupling speed, completeness, and impurity risk.
- Protecting group strategy affects how aggressively the peptide must be treated during synthesis and release.
A peptide doesn’t become high quality because one step went well. It becomes high quality when the support, linker, chemistry, and purification plan all point in the same direction.
This is one reason difficult peptides require experience. When a sequence misbehaves, the answer isn’t always “use more reagent.” Sometimes the problem started with the wrong solid support, the wrong cleavage logic, or a reagent set that pushed the chemistry toward side products instead of clean conversion.
The SPPS Workflow From Manual Synthesis to Automation
Manual solid phase peptide synthesis teaches you respect for process. Every wash, every deprotection, every transfer matters because small inconsistencies can propagate through the entire sequence.

What manual synthesis teaches you
In a manual workflow, a chemist swells the resin, performs each deprotection step, adds activated amino acids, washes thoroughly, and monitors the chain’s progress by direct observation and testing. This approach offers flexibility. It’s useful when developing a method, troubleshooting a difficult sequence, or working on a small set of unusual targets.
It also exposes where variability enters. Inconsistent timing, incomplete solvent exchange, poor mixing, and handling differences between operators can all alter crude quality. Manual synthesis can absolutely work well, but it depends heavily on disciplined execution.
For labs thinking about process design beyond a fully manual setup, this guide for manufacturing semi-automation gives a useful framework for understanding how partial automation can reduce repetitive handling errors without removing process flexibility.
Why automation improves reproducibility
Automation matters because peptide synthesis is repetitive chemistry. That’s exactly the kind of workflow instruments handle well. Automated synthesizers meter reagents consistently, apply programmed reaction times, and standardize washes across every cycle.
That consistency is now a real differentiator for research supply. A Chemputer-based automation report describes modern automated SPPS systems that can deliver 3x throughput compared to manual methods and achieve over 90% consistency in yield, with reduced failure rates for complex sequences.
Those numbers aren’t just operational bragging points. They connect directly to what a buyer cares about:
- Batch reproducibility means one lot behaves more like the next.
- Controlled reagent delivery reduces cycle-to-cycle drift.
- Reliable washing lowers the chance that carryover creates side products.
- Scalable execution makes it easier to produce multiple research lots with similar quality profiles.
A short visual overview helps if you want to see how automated lab workflows map onto the chemistry:
Automation doesn’t eliminate the need for expertise. Someone still has to choose the right chemistry, resin, linker, and purification strategy. What it does remove is avoidable variation in repetitive steps, and that’s a major reason well-run automated workflows support more reliable peptide production.
Ensuring Purity and Identity Purification and Analysis
A peptide coming off the resin isn’t automatically ready for research. At that stage, it’s usually still mixed with protecting group remnants, truncated sequences, deletion products, and cleavage-related impurities. The molecule exists, but the sample is not yet analytically trustworthy.
Cleavage is where protected chemistry becomes a real sample
The first job after chain assembly is cleavage. This step detaches the peptide from the resin and removes the relevant protecting groups so the intended sequence exists in its usable form.
That transition is chemically decisive. If cleavage conditions are poorly matched to the sequence, you can damage sensitive residues, leave partial protection behind, or generate new impurities that weren’t present during chain assembly.
After cleavage, chemists isolate the crude peptide and evaluate how much cleanup will be required. For straightforward sequences, the crude may already look manageable. For difficult sequences, especially those with incomplete couplings or aggregation-related issues earlier in synthesis, the crude can contain a dense mixture of near-neighbor impurities.

HPLC and MS are what make a COA meaningful
HPLC plays a central role. High-performance liquid chromatography separates the target peptide from related impurities based on how each component behaves in the chromatographic system. In plain terms, it’s how a supplier moves from “we made the sequence” to “we isolated the right material cleanly.”
If you’ve ever wondered why purity claims deserve scrutiny, this is the answer. The stated purity isn’t a marketing adjective. It reflects how effectively the purification process separated the desired peptide from failed sequences and side products.
A second analytical tool completes the picture. Mass spectrometry verifies molecular identity by confirming the expected mass of the product. It doesn’t replace HPLC, and HPLC doesn’t replace MS. One tells you how clean the sample is relative to other components. The other helps confirm that the main component is the peptide you intended to make.
A COA is only useful when it reflects real purification and real analytical verification. Without that, a purity number is just a label.
When you read a Certificate of Analysis, think like a chemist:
- Purity statement asks how much of the sample is the main peptide peak under the stated method.
- Mass confirmation asks whether the principal component matches the expected identity.
- Lot-specific documentation asks whether the data belongs to that batch, not a generic template.
- Supporting analytical records ask whether the supplier is transparent enough to let you evaluate the material credibly.
For research use, this matters more than branding language. A peptide with convincing analytical support is easier to trust in assay development, method validation, and routine lab work. A peptide without that support creates uncertainty before the experiment even begins.
Troubleshooting and Sourcing High-Quality Peptides
Peptide synthesis looks elegant in diagrams. At the bench, it can be stubborn. Sequences fold on resin, couplings stall, protecting groups don’t come off cleanly, and a peptide that seemed simple on paper can become difficult after only a few residues.
Where syntheses commonly go wrong
One of the biggest troublemakers is aggregation. Some growing peptide chains begin to interact with themselves while still attached to the resin. When that happens, reagents can no longer access every reactive site evenly, and the synthesis starts losing efficiency in ways that are hard to reverse.
This is not a niche problem. Sigma-Aldrich’s technical discussion of overcoming aggregation in SPPS notes that peptide aggregation affects over 50% of long-sequence syntheses and can increase costs by 2-5 times because of failed runs and lower yields. It also notes that chemical additives are often more effective than changing the resin type alone.
That matters for buyers because difficult synthesis histories leave fingerprints on the final product. A supplier may still recover the target peptide, but if the process struggled repeatedly, purification becomes more demanding and lot-to-lot consistency can become harder to maintain.
Common practical failure points include:
- Incomplete deprotection that leaves unreactive sites on part of the growing chain
- Weak coupling performance that creates truncated products
- Aggregation on resin that blocks reagent access
- Cleavage-related damage that alters sensitive sequences
- Insufficient purification that leaves near-neighbor impurities in the final lot
Some peptide problems are sequence problems, not operator mistakes. Good manufacturers recognize the difference and adapt the chemistry instead of forcing the same default protocol onto every target.
How to judge a peptide supplier like a chemist
Once you understand the synthesis pathway, sourcing gets easier. You stop asking only “What peptide is this?” and start asking “What process discipline likely produced this batch?”
Look for signs that the supplier understands the chemistry behind the label.
- Transparent COAs: A serious supplier provides lot-specific Certificates of Analysis rather than generic summaries.
- Identity support: Mass confirmation should be part of the analytical story, not an afterthought.
- Purity disclosure: Stated purity should be clear and tied to actual batch documentation.
- Manufacturing clarity: A transparent supply chain and clear production standards reduce guesswork.
- Research-use positioning: The company should clearly present products for laboratory, analytical, or preclinical use, not blur compliance boundaries.
If you’re comparing vendors for peptides such as BPC-157, TB-500, GHK-Cu, PT-141, Selank, or Semax, don’t focus only on the product name. The same sequence can arrive as a very different research material depending on synthesis control, purification quality, and documentation standards.
A chemist doesn’t read peptide sourcing as a shopping exercise. They read it as a chain of evidence. Was the sequence built under the right conditions? Was the crude cleaned up properly? Was identity confirmed? Is the documentation specific enough to trust the lot in actual research?
That’s the practical value of understanding solid phase peptide synthesis. It makes you a harder customer to mislead and a better scientist when you evaluate incoming materials.
If you’re looking for research peptides supported by transparent batch documentation, USA-based manufacturing, and stated purity levels up to 99.5%, learn more at Peptide Warehouse USA and explore options for laboratory, analytical, and preclinical research.






Leave a comment