What Is Peptide Therapy: 2026 Research and Science Guide
Most explanations of peptide therapy skip the part that matters most. Are we talking about approved medicines used in clinical care, or about research peptides sold for laboratory work? If you don’t separate those two worlds, the whole topic gets muddy fast.
That distinction matters because peptide therapy sits at the intersection of real clinical progress, active drug development, and a large research supply market. Interest has risen sharply. Google Trends data cited by OpenLoop Health shows a +300% increase in searches for “cost of peptide therapy” and an 80% increase in general “peptides” searches in the United States between April 2025 and April 2026.
If you’re trying to understand what is peptide therapy, the useful starting point isn’t hype. It’s mechanism, evidence, and quality control. Once those pieces are clear, the difference between a clinically validated peptide drug and a research compound becomes much easier to see.
Table of Contents
- Understanding Peptides and Peptide Therapy
- The Cellular Signaling Mechanism of Peptides
- Common Categories of Research Peptides and Their Applications
- Evaluating the Evidence Behind Peptide Benefits
- Administration Routes and Safety Protocols for Research
- A Researcher’s Guide to Navigating Quality and Sourcing
- Conclusion The Future of Targeted Biological Intervention
Understanding Peptides and Peptide Therapy
What peptides are at the molecular level
Peptides are short chains of amino acids. You can think of amino acids as molecular letters, and peptides as short messages written with those letters. Proteins are built from the same alphabet, but they’re generally longer and fold into larger, more complex structures.
A simple analogy helps. If a protein is a full instruction manual, a peptide is often more like a text message. Short, specific, and aimed at one task.

Some peptides occur naturally in the body and act as signaling molecules. Others are made synthetically to mimic or modify those signals.
What peptide therapy means in practice
So what is peptide therapy? In plain language, it’s the use of selected peptides to influence biological processes through cell signaling. The idea isn’t to overwhelm the body. It’s to send a message that a cell already knows how to interpret.
That sounds straightforward, but readers often get tripped up here because the phrase covers very different realities:
- Clinically used peptide drugs are studied in human trials, approved in specific settings, and manufactured under strict standards.
- Research peptides are used in laboratory, analytical, or preclinical settings and are not the same thing as approved therapies.
- Marketing language often blurs those categories, which makes weak evidence sound stronger than it is.
Peptide therapy isn’t one product class with one level of evidence. It’s a broad biological strategy applied across very different regulatory contexts.
That helps explain the current attention. Some peptide medicines are now central to mainstream care, while many other compounds remain exploratory. Understanding that split is more important than memorizing a long list of peptide names.
The Cellular Signaling Mechanism of Peptides
How can a short chain of amino acids change what a cell does? The answer is usually not brute force chemistry. It is selective signaling.
Receptors are the key concept
The main mechanism is receptor-mediated signaling. A peptide reaches a cell, binds a compatible receptor on the cell surface or in its local environment, and that binding changes the receptor’s shape or activity. That change starts an intracellular signaling cascade, which can alter gene expression, secretion, metabolism, migration, or growth behavior.
A receptor works like a molecular sensor. It does not respond to any peptide that happens to drift by. It responds to peptides with the right sequence, shape, charge distribution, and binding affinity. This is why small changes in peptide structure can produce very different biological effects.
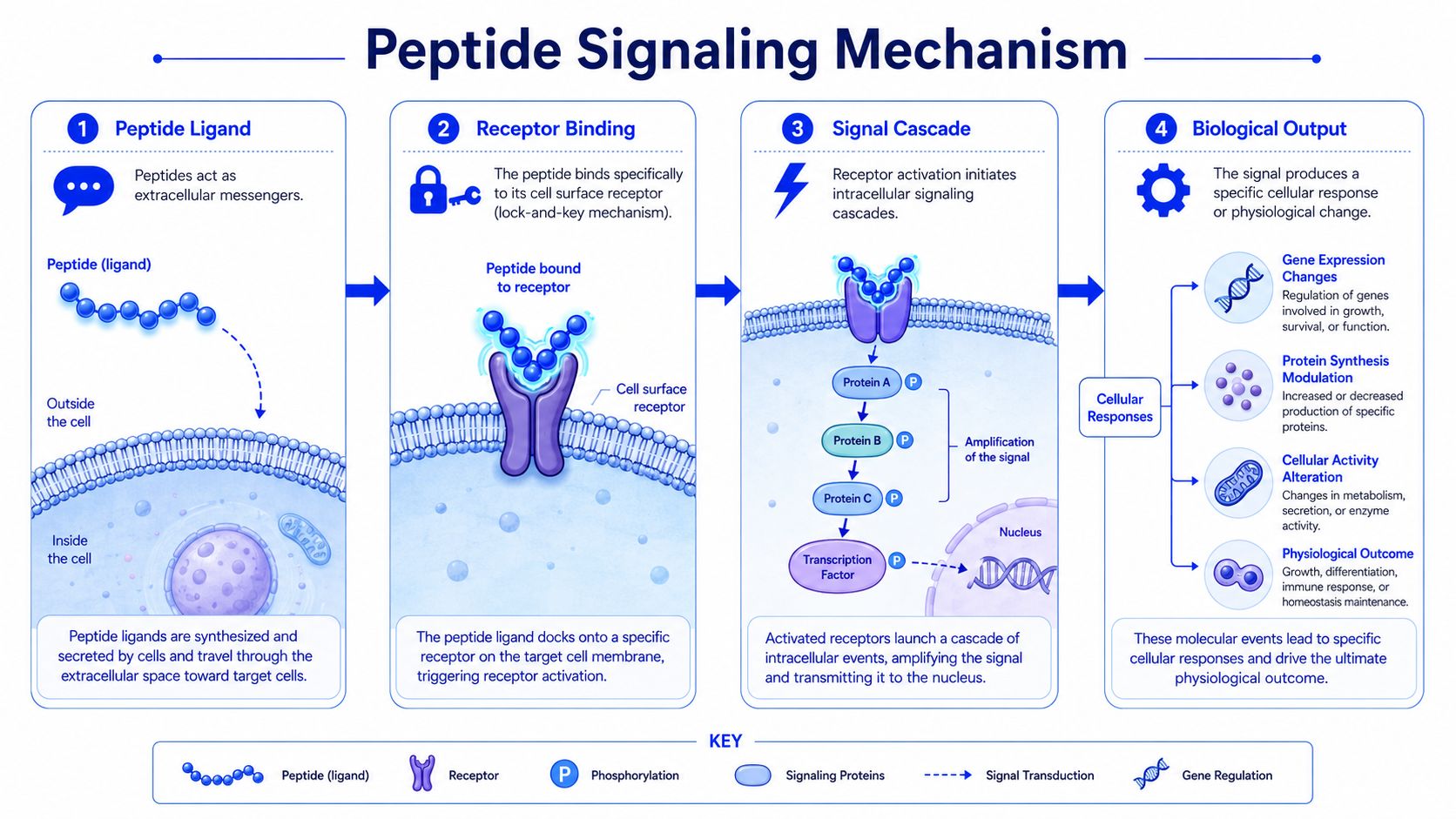
A simplified sequence looks like this:
- A peptide enters the system and remains intact long enough to reach potential targets.
- It encounters a compatible receptor on a target cell or tissue.
- Binding alters receptor activity, which initiates downstream signaling inside the cell.
- The cell changes its behavior according to that signal and the surrounding biological context.
That last point causes a lot of confusion. Peptides do not carry a universal instruction like “repair tissue” or “burn fat.” They trigger responses only in cells that express the relevant receptor, and the result depends on what signaling machinery those cells already have available. The same signal can produce different outcomes in different tissues.
Why peptide signaling can appear more targeted
Many small-molecule drugs are designed to inhibit an enzyme, block a channel, or dampen a pathway. Peptides often act more like endogenous messages that the body already uses for coordination. The distinction matters because it changes how researchers think about mechanism. The question is often not “What process does this shut down?” but “Which receptor does this activate, in which cells, under which conditions?”
That does not make peptides automatically safer or more predictable. Receptors can be expressed in multiple tissues. Signaling pathways can cross-talk. Dose, timing, degradation, and receptor desensitization all affect the result.
A useful comparison is below:
| Feature | Peptide signaling view | Broad inhibition view |
|---|---|---|
| Primary action | Mimics or modifies a biological message | Suppresses a protein, enzyme, or pathway |
| Target logic | Often receptor-dependent | Often enzyme- or pathway-dependent |
| Biological effect | Shaped by receptor distribution and cell state | Shaped by the blocked process |
Practical rule for researchers: If a peptide claim does not clearly identify the receptor, the downstream pathway, and the experimental context, the claim is not specific enough to evaluate.
This is also where the split between approved peptide medicines and research peptides becomes important. For established clinical drugs, receptor targets, pharmacology, manufacturing controls, and safety data are usually characterized in far greater detail. In the research peptide world, those elements may be incomplete, inferred from preclinical work, or described inconsistently by vendors.
That is why mechanism should never be separated from documentation. If a supplier claims a peptide has a defined signaling action, a careful researcher should ask for evidence that the material itself is what the label says it is. Sequence confirmation, purity testing, impurity profiling, and a readable certificate of analysis matter because signaling data are only as credible as the compound being tested. Without that foundation, “mechanism” can become a story attached to an uncertain sample.
Common Categories of Research Peptides and Their Applications
How should a researcher sort a peptide field that is often presented as a single trend? The most useful approach is to classify peptides by the biological system they are meant to influence, then ask whether the evidence comes from approved clinical use, human studies, animal models, or cell-based experiments.
That distinction matters because “peptide therapy” is not one thing. Some peptides are established medicines with defined indications. Others circulate mainly in research settings, where mechanism, material quality, and vendor documentation can vary widely. Grouping them together hides the difference between a regulated drug and an experimental reagent.
A practical mental model helps here. Categories are like folders in a lab notebook, not verdicts about efficacy. They organize questions. They do not prove outcomes.
Healing and repair research
Peptides such as BPC-157 and TB-500 are often placed in a repair-focused category because researchers discuss them in relation to tissue response after injury. Depending on the model, that discussion may include cell migration, inflammatory signaling, angiogenesis, and wound-healing processes in tissues such as muscle, tendon, ligament, or gastrointestinal lining.
The key point is scope. These are research applications and mechanistic hypotheses, not a blanket statement that the peptides are established treatments for human repair.
This category also shows why sourcing documents matter. If a lab is studying a peptide for effects on tissue closure or inflammatory markers, the interpretation depends on knowing the sample identity, purity, and possible impurities. A result attached to a poorly characterized vial is hard to trust, even if the biological story sounds plausible.
Metabolic and weight management research
This category sits closest to routine clinical medicine because some peptide-based drugs that affect appetite and glucose regulation are already approved for defined indications. Examples commonly discussed in research and clinical contexts include semaglutide and tirzepatide, while retatrutide is often described in the investigational setting.
Mechanistically, these compounds are easier to classify than many “wellness” peptides. The research question usually centers on receptor-mediated signaling related to appetite, gastric emptying, insulin response, energy balance, or body weight. That gives investigators clearer endpoints and a more direct path from mechanism to measurable outcomes.
Even here, categories need care. A peptide can belong to a clinically mature class while a specific molecule within that class remains experimental.
Nootropic and neurobiology research
Peptides such as Selank and Semax are commonly discussed in neurobiology and nootropic research. The useful scientific question is not whether they are “brain boosters.” It is which neural systems are being tested, which endpoints are being measured, and whether those endpoints are objective.
Neurobiology is especially easy to overstate because many outcomes sit close to perception, behavior, or self-report. In one study, a peptide might alter a biochemical marker or an animal behavior. That is not the same as demonstrating a reliable human cognitive benefit in a controlled clinical setting.
For this category, researchers should read claims with extra discipline. Ask what species was studied, what assay was used, and whether the proposed mechanism matches the observed effect.
Skin and cosmetic biology research
GHK-Cu is often grouped under skin and cosmetic biology because researchers connect it to dermal remodeling, collagen-related processes, and visible skin outcomes. The attraction of this category is obvious. Surface-level changes are easy to market.
The science is more nuanced. A peptide may affect pathways involved in extracellular matrix turnover or skin signaling without establishing a proven therapeutic effect for a specific medical indication. Mechanistic interest and clinical proof are different standards.
This is another area where the line between research peptide discussion and product marketing often blurs. For a careful reader, that is a signal to check the study type and the quality record of the material used.
Here is a simple reference map:
| Category | Example Peptides | Primary Area of Research |
|---|---|---|
| Healing and repair | BPC-157, TB-500 | Tissue response, migration, inflammatory signaling, wound-healing models |
| Metabolic | Semaglutide, tirzepatide, retatrutide | Appetite signaling, metabolic regulation, weight-related endpoints |
| Nootropic | Selank, Semax | Neurobiology, signaling, cognitive and behavioral models |
| Skin and cosmetic biology | GHK-Cu | Dermal remodeling, collagen-related processes, cosmetic biology |
A useful screening method is to ask three questions before accepting any application claim:
- What endpoint was measured? Receptor activation, tissue closure, body weight, behavioral output, histology, or something else.
- What model produced the result? Cell culture, animal study, human trial, or post-market clinical use.
- What documentation supports the material itself? Sequence identity, purity data, and a readable COA should match the peptide being discussed.
A peptide can look impressive in a tightly controlled mechanistic model and still remain uncertain for real-world human use. That gap is where careful classification, evidence grading, and sourcing review become part of the same scientific task.
Evaluating the Evidence Behind Peptide Benefits

How much confidence should you place in a claimed peptide benefit?
The answer depends less on the peptide name than on the type of evidence behind it. In peptide science, one label can cover very different situations: an approved drug with human outcome data, an early candidate with animal results, or a research compound discussed mainly through mechanistic papers and vendor pages. If you do not separate those categories, the discussion gets muddy fast.
Established clinical evidence and emerging research
Start with the central distinction. Clinical evidence asks what happened in people under defined study conditions. Preclinical evidence asks whether a biological effect appears plausible or measurable in cell systems, animal models, or analytical experiments. Those are related questions, but they are not the same question.
A useful analogy is a map versus a road test. Preclinical work can show where a pathway exists and which turns look promising. Human trials show whether the vehicle reaches the destination, under real constraints, with acceptable safety and reproducibility.
That distinction matters because peptide discussions often place approved therapeutics and research-only compounds in the same conversational bucket. Scientifically, that is too broad. Some peptide classes already support routine clinical use for specific indications. Other compounds remain exploratory, with interesting signals but limited human evidence. A reader trying to understand “peptide therapy” needs to know which side of that line a given claim belongs on.
As noted earlier, the peptide field includes both approved medicines and a large development pipeline. The strongest clinical visibility today comes from metabolic medicine, where peptide-based drugs have produced measurable human outcomes and regulatory decisions. That level of evidence is very different from a repair, neuroactive, or performance-related peptide discussed mainly in rodent models or cell assays.
A short explainer is useful here:
How to read peptide claims critically
A practical way to evaluate a peptide claim is to place it on an evidence ladder.
-
Mechanistic rationale
The peptide interacts with a receptor, enzyme, or signaling pathway that makes biological sense. -
Preclinical evidence
Cell or animal studies show a measurable effect under controlled conditions. -
Human clinical evidence
Investigators test the peptide in people and report defined endpoints such as body weight, symptom scores, lab values, or adverse events. -
Regulated therapeutic use
A specific peptide product meets regulatory standards for manufacturing, quality control, and clinical evidence in a defined indication.
Confusion usually starts when readers treat the first rung as if it were the fourth. A plausible mechanism is useful. It is not proof of clinical benefit. An animal signal is worth studying. It does not establish effectiveness in humans.
This is also where the research-versus-clinic distinction becomes practical rather than semantic. If a peptide is discussed in research settings, the question is not only “What effect was reported?” It is also “What material was tested, how was it characterized, and does the documentation support the claim?” For research peptides, sequence confirmation, purity testing, batch consistency, and a readable certificate of analysis matter because weak material can create weak conclusions.
So the right question is not whether peptides “work” in the abstract. The better question is narrower and more scientific: Which peptide, for what endpoint, in which model, with what quality controls, and with what level of human evidence?
That framing helps separate established clinical facts from active but unsettled areas of research.
Administration Routes and Safety Protocols for Research
Why route of administration matters
A lot of readers assume a peptide can be handled like a standard oral compound. Often, it can’t. Many peptides have poor oral bioavailability, which means the digestive system and related processes can break them down before they reach the intended target in usable form.
That is why subcutaneous and intramuscular administration show up so often in peptide work. In some research settings, other delivery approaches are studied as well, including nasal formats, but route selection always comes back to stability, absorption, and the question being tested.

The formulation side is just as important. Analytical workflows used in peptide development include LC-MS/MS, HPLC or UHPLC, and conformational methods such as CD and NMR because structure, purity, and stability directly affect behavior.
Research handling and protocol discipline
For research settings, administration is only one part of the protocol. Handling standards matter just as much.
Key points include:
- Match route to purpose. A route used in one experimental design may be inappropriate for another.
- Protect sample integrity. Storage, reconstitution, and handling errors can alter results before a study even begins.
- Track batch identity. If one lot differs from another, downstream findings may not be comparable.
- Separate research from personal use. Research peptides are sold for laboratory, analytical, or preclinical work. They are not consumer self-treatment tools.
The timeline question also deserves realism. treatment timelines and outcomes vary substantially by peptide, protocol, and individual biological factors. Some studies report measurable changes within weeks, while others require months for fuller effects in research models.
Don’t treat a peptide timeline like a fixed clock. Response depends on the molecule, the formulation, the protocol, and the biology under study.
That variability is one reason standardized protocols are so important. Without them, it becomes hard to tell whether a disappointing result reflects the biology, the material, or the method.
A Researcher’s Guide to Navigating Quality and Sourcing
A peptide can be chemically interesting and still be useless for serious research if the sourcing is poor. Many public discussions fail in this regard. They explain potential effects but say almost nothing about how a lab should judge material quality.
What a good Certificate of Analysis should tell you
A Certificate of Analysis, or COA, is not just a marketing attachment. It is part of the chain of evidence showing what a batch is, how it was tested, and whether its stated purity and identity are supported.
According to Renew Integrative Health’s discussion of the sourcing gap in peptide content, a critical gap exists between legitimate research supply chains and unregulated online vendors. The same source highlights quality markers researchers should verify, including batch testing, purity verification up to 99.5%, and traceability documented in a Certificate of Analysis.
A useful COA review checklist includes:
- Identity confirmation through appropriate analytical methods
- Purity reporting with a clear stated value
- Batch traceability so the lot can be linked to documentation
- Microbial and endotoxin reporting where relevant to the application
- Test dates and lot matching so the document corresponds to the material in hand
How to think about supplier credibility
Researchers should be skeptical of suppliers that provide polished claims but thin documentation. A peptide listing is not proof of quality.
Look for suppliers that make verification possible, not just easy to assume. Useful questions include:
| Question | Why it matters |
|---|---|
| Is there lot-specific documentation? | Generic documents don’t confirm the batch you received |
| Is purity stated clearly? | Vague quality language isn’t analytically useful |
| Are microbial or endotoxin reports available when relevant? | Contaminants can distort results and compromise handling |
| Is traceability built into the record? | Reproducibility depends on it |
Good peptide sourcing protects research integrity before it protects convenience.
That matters even more in a field where public interest runs ahead of public understanding. If the compound is poorly characterized, every downstream interpretation becomes weaker.
Conclusion The Future of Targeted Biological Intervention
Peptide therapy makes sense once you view peptides as signaling tools rather than magic compounds. They are short amino acid chains that can influence biology through receptor-mediated communication, and in some areas, especially metabolic medicine, that approach has already produced major clinical progress.
At the same time, the field of peptides comprises far more than approved drugs. Many compounds remain preclinical, research-only, or early in development. That’s why the most important habit in this field is separation. Separate mechanism from marketing, clinical evidence from exploratory data, and legitimate sourcing from vague online claims.
If you’re evaluating peptides for laboratory or analytical work, quality documentation isn’t optional. It’s the basis for reproducible results.
If you’re looking for high-purity peptides for laboratory, analytical, or preclinical applications, Peptide Warehouse USA offers USA-made research compounds with lot-specific documentation, including COAs, microbial and endotoxin reports, and stated purity levels up to 99.5%. Learn more and explore options that support careful, evidence-minded research.






Leave a comment